Robotic beating-heart totally endoscopic coronary artery bypass
Clinical vignette
Since the mid-nineties, minimally invasive direct coronary artery bypass grafting (MIDCAB) has evolved thanks to technological development of robotically assisted coronary surgery known as totally endoscopic coronary artery bypass (TECAB). Here, we report the case of a 51-year old lady with typical angina pectoris without any other relevant comorbidity. The angiogram showed a single and severe lesion at the ostium of the left anterior descending (LAD) coronary artery. Following an interdisciplinary heart team approach, a TECAB was proposed and accepted by the patient. This tutorial demonstrates the technical aspects of a single beating-heart TECAB.
Surgical techniques
Preparation
After standard induction of anesthesia and double lumen intubation for single right lung ventilation, the patient lies in a supine position on a 15-cm diameter roll placed parallel to the left in his back with the table tilted 15° to the right side.
Exposition
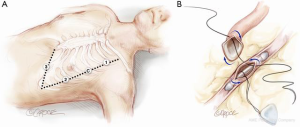
The camera port is inserted into the left 4th intercostal space, at the level of the anterior axillary line. After connection of the CO2 insufflation (3–10 mmHg pressure), the 3D-camera allows left chest cavity exploration especially for positioning the right and left arms (1 and #2 respectively) through the 2nd and 6th intercostal spaces, where trocars are inserted under visual control. The endoscopic stabilizer (arm #3) is placed later in an additional port, at half distance between the xyphoid appendage and the left anterior axillary line (Figure 1A).
Operation
After orientation in the thorax, a careful pericardiotomy is performed to localize the target vessel. Then the internal mammary artery (IMA) is harvested in a skeletonized or pedicled manner based on the surgeon’s preference. Once the correct coronary artery target is identified (usually the LAD artery), the endostabilizer is slowly placed in order to have a clear target zone. Particular caution should be taken for epicardial dissection of the target vessel. A temporary silastic snare loop is placed proximally around the vessel in order to control the bleeding during the arteriotomy. A 5 to 7 mm arteriotomy is made, a coronary shunt (Medtronic, Minneapolis, MN, USA) is inserted, and the silastic snare loop is then released. The anastomosis is performed with a running suture using a single needle 8/0 expanded polytetrafluoroethylene (ePTFE) of 15 cm length (Figure 1B).
Here are listed some considerations for double/multiple bypasses. Initial settings up to the trocars’ placement are similar. If a T-/Y-graft is needed, the right IMA (RIMA) is then harvested first in order to avoid damage to an already hanging left IMA (LIMA). The latero-terminal anastomosis with the RIMA on the LIMA is performed first with a 7–10 cm, 8/0 ePTFE running suture. After completion of the LIMA-to-LAD anastomosis as described previously, the endostabilizer is removed and the trocars disconnected from the robotic arms. The table is tilted to 30° (additional 15°) to the right in order to shift the heart medially. After reconnection of the arms to the trocars, the endostabilizer is repositioned and locked in the new target zone. Finally the suture is performed in the same manner. A femoral-femoral bypass (beating-heart assisted) may be useful in some patients, in order to have a better vision of the left ventricular lateral wall.
In the case of a sequential bypass, the endostabilizer will only have to be displaced from one target to the other, and the second anastomosis (mainly on the diagonal artery) performed in a conventional way. No repositioning of the robot is needed.
Completion
After hemostatic control of the suture, all the tools and needles are removed from the thoracic cavity and a chest tube is inserted through the inferior incision. The patient was extubated and transferred to the intensive care unit for 24 hours and eventually discharged home on the fourth postoperative day.
Comments
Clinical results
Since the first report (1) of TECAB at the turn of the millennium, several teams have adopted and unfortunately also abandoned the technique. Only a few motivated and convinced early adopters have remained active and further developed its use. Beating-heart totally endoscopic bypass surgery does offer a valid alternative to conventional revascularization strategies. Indeed perioperative outcomes such as myocardial infarction, stroke, acute kidney injury, bleeding, atrial fibrillation and mortality were comparable to conventional surgery (2).
Advantages
Since the chest remains closed by avoiding the sternotomy, much-feared sternal dehiscence and infection are no longer a consideration. While generally minimizing surgical trauma, it also hastens the patient’s recovery and therefore has a positive impact on the patient’s early return to work or normal daily activities (3).
Caveats
Although robotics has allowed safe, effective and reproducible single LIMA-to-LAD coronary bypass (4), its place in the coronary disease therapeutic armamentarium has still to be more broadly accepted. First, hybrid coronary revascularization will most probably expand further because endovascular treatments are effective and newer generation of (bioresorbable) stents will improve even more those positive results. Second, the arterial bypass LIMA-to-LAD still is an undeniable survival predictor in coronary disease (5). Because in only a few years from now, many patients and cardiologists will not accept conventional CABG for a single bypass, this surgical therapy must be proposed in minimally invasive techniques.
Acknowledgements
We thank our whole medical and nursing team for their efforts. This work was supported in part by an internal grant of the Mont-Godinne Foundation to L Melly.
Footnote
Conflicts of Interest: JL Jansens is a Consultant for Intuitive Surgical. The other authors have no conflicts of interest to declare.
References
- Loulmet D, Carpentier A, d'Attellis N, et al. Endoscopic coronary artery bypass grafting with the aid of robotic assisted instruments. J Thorac Cardiovasc Surg 1999;118:4-10. [Crossref] [PubMed]
- Cao C, Indraratna P, Doyle M, et al. A systematic review on robotic coronary artery bypass graft surgery. Ann Cardiothorac Surg 2016;5:530-43. [Crossref] [PubMed]
- Zaouter C, Imbault J, Labrousse L, et al. Association of Robotic Totally Endoscopic Coronary Artery Bypass Graft Surgery Associated With a Preliminary Cardiac Enhanced Recovery After Surgery Program: A Retrospective Analysis. J Cardiothorac Vasc Anesth 2015;29:1489-97. [Crossref] [PubMed]
- Bonaros N, Schachner T, Lehr E, et al. Five hundred cases of robotic totally endoscopic coronary artery bypass grafting: predictors of success and safety. Ann Thorac Surg 2013;95:803-12. [Crossref] [PubMed]
- Loop FD, Lytle BW, Cosgrove DM, et al. Influence of the internal-mammary-artery graft on 10-year survival and other cardiac events. N Engl J Med 1986;314:1-6. [Crossref] [PubMed]





