Type I and Type II hybrid aortic arch replacement: postoperative and mid-term outcome analysis
Introduction
The management of aortic arch aneurysms remains a clinical challenge. Open total arch procedures require the use of complex circulatory management and adjunct cerebral protection strategies. Given the complexity of the procedure, patients with multiple comorbidities may experience significant morbidity and mortality from both neurological and cardiovascular complications (1-3). In these high risk patients, alternative therapies are sought. The introduction and regulatory approval of thoracic endovascular repair (TEVAR) for descending aortic pathology has seen its application for many off-label uses, including aortic dissection (4,5), traumatic transection (6), infection/fistula and penetrating atherosclerotic ulcers (7,8). This application has been extended to the use of TEVAR for the stenting of the aortic arch with open great vessel debranching. The “hybrid” aortic arch repair, with debranching and reimplantation, or bypass of aortic arch vessels and TEVAR with endografting of the aortic arch, is emerging as the ideal treatment option for complicated aortic arch pathologies (9-11), especially in patients with severe comorbid status.
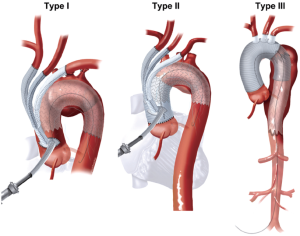
The hybrid arch repair seeks to limit operative, bypass, and circulatory arrest times by simplifying and shortening the arch repair procedure. The principle concept entails reimplantation or bypass of aortic arch vessels, and TEVAR implantation landing proximally in “zone 0” (ascending aorta-innominate origin) which is either suitable for use as a landing zone natively, or has been replaced with a Dacron graft to construct an artificial landing zone (Figure 1). Multiple studies have demonstrated the feasibility of this approach to the aortic arch, highlighting acceptable mortality and morbidity (9-11). We report our institutional experience with the arch hybrid operation, with midterm follow-up on these patients.
Methods
From 2005 to present, 685 patients have received thoracic aortic endovascular repair at our institution. Of these, 104 received TEVAR with proximal aortic repair: 57 ascending/hemiarch replacements with antegrade stenting of the descending aorta for DeBakey Type I dissection, and 47 hybrid arch debranching procedures (Figure 2). The variations of these repairs can be classified as Types I, II, and III.
Type I hybrid arch
Of the 47 patients, 28 received a Type I hybrid arch repair, where there is an isolated arch aneurysm with good native landing zones (zone 0 and zone 3/4). Type I repair consists of reimplantation of the aortic arch vessels using a four-branched Dacron graft, which is sewn to the native ascending aorta just above the sinotubular junction (Figure 1). This can be done without cardiopulmonary bypass in hemodynamically stable patients who do not possess a calcified aorta, using a side-biting clamp; otherwise it is done on cardiopulmonary bypass with or without a short aortic cross-clamp time.
If there is concern that the exposure of the left subclavian artery will be difficult via a median sternotomy, a pre-emptive left subclavian to left common carotid bypass is an option, and only innominate and left common carotid arteries are then reimplanted to the ascending aorta. After the arch debranching is complete, stent grafts can then be deployed via the fourth limb of the graft into the aortic arch. In some patients endograft implantation is delayed and deployed later during the same hospital stay via a retrograde iliofemoral approach. If carotid-subclavian bypass is performed, it is critical that the proximal left subclavian artery is ligated or coiled in order to prevent a Type II (branch vessel) endoleak upon aortic stenting.
Type II hybrid arch
8 patients received Type II hybrid arch repair. This is performed in patients with proximal extension of aneurismal pathology into the ascending aorta, making the native aorta unsuitable for zone 0 stent graft landing. This can be addressed through replacement of the ascending aorta, creating a Dacron ascending zone 0. At our institution, ascending aorta replacement, or a Type II hybrid arch solution, is done for zone 0 diameter >3.7 cm, as we and others noted an increased risk of retrograde Type A dissection in the deployment of proximal stent grafts with diameter greater than 4 cm. Type II arch hybrid operations require cardiopulmonary bypass and sometimes brief circulatory arrest for proximal aortic reconstruction, which can be done using either retrograde or selective antegrade perfusion strategies. After arch debranching and reconstruction of zone 0 with an ascending aortic graft, the endograft can be deployed through the arch with a Dacron zone 0 landing zone.
Type III hybrid arch
11 patients received Type III hybrid arch repairs, which encompasses a total arch replacement with descending elephant trunk, combined with delayed second stage retrograde TEVAR repair, for extensive aortic pathologies such as “mega-aorta” syndrome. Type III hybrid arch repairs are not classic hybrid arch repair procedures. All patients received either selective antegrade perfusion or a combination of antegrade and retrograde cerebral perfusion during the circulatory arrest period. These patients are not included in further analysis due to significant differences in patient population, disease process, and most importantly, operative technique, as the TEVAR solution is beyond the zone 0 proximal extension.
In all cases, the patient was deemed unsuitable for traditional open repair due to age or other comorbidities. Patients were preoperatively evaluated with multi-slice computed tomography angiogram (CTA). CTA images were processed into three-dimensional images by an outside service (M2S, West Lebanon, NH) for preoperative planning and proper sizing of endografts deployed in the arch. Surgery was performed in the hybrid operating room equipped with universal floor-mounted angiographic C-arm system for fluoroscopic guidance during and after endograft deployment.
Intraoperative transesophageal echocardiography (TEE) was performed in all cases. All patients received at least one form of intraoperative neuromonitoring: intraoperative electroencephalogram (EEG), somatosensory-evoked potentials (SSEPS), and when available, cerebral oximetry using near-infrared spectroscopy (NIRS).
Statistical analysis and follow-up methods
All statistical comparisons were done using SPSS 19 (SPSS Inc., Chicago, Il). Categorical variables were compared using Fisher’s exact test. Continuous variables were compared using Student’s t-test. Long-term survival was monitored through the national Social Security Death Index (SSDI) and cumulative survival was calculated using Kaplan-Meier analysis. All patients were followed postoperatively, with regular contrast CTA imaging (discharge, 30 day, six month, and yearly). Follow-up was 100% in all groups. Institutional review board approval was obtained.
Results
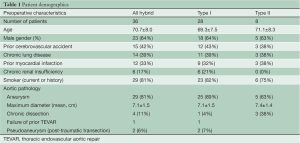
From 2005 to present, 36 Types I and II hybrid arch operations were performed at the Hospital of the University of Pennsylvania. Mean age was 70.7±8 years, 23 (64%) were male (demographics is presented in Table 1). 42% of patients had prior stroke, 33% had previous myocardial infarction, and 81% had some history of smoking. 29 (81%) of these presented with aneurysmal disease of the aortic arch (mean aortic diameter =7.1±1.5 cm). 4 presented with chronic aortic dissection, 2 presented with pseudoaneurysm of the aortic arch secondary to remote traumatic transection, and 1 patient presented with proximal degeneration in the setting of prior TEVAR repair (for descending aortic aneurysm) (Table 2). Prior surgical history included 3 previous CABG, 1 patent foramen ovale repair, 1 Type A dissection repair via sternotomy approach, and one thoracoabdominal aortic aneurysm repair. 2 patients had prior thoracic aortic endograft repairs, one who presented with proximal degeneration of the prior repair, and one who presented with a new unrelated saccular aneurysm in the mid arch.
Full table
Full table
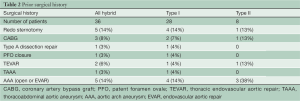
Of the 28 Type I hybrid arch repairs, 16 (57%) were done off cardiopulmonary bypass, using a side-biting clamp on the ascending aorta for great vessel debranching (Table 3). Twelve were done on cardiopulmonary bypass (mean time of 193±58 minutes) with aortic cross clamp (mean 44±27 minutes). Eighteen (64%) patients received TEVAR antegrade through the fourth limb of the aortic graft, 7 (25%) patients received concomitant retrograde TEVAR via iliofemoral approach. Three (11%) patients did not receive endografting during the same operation, but received afterwards during the same hospital stay via iliofemoral access (Table 3).
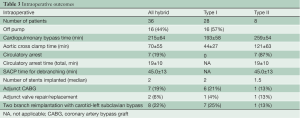
Full table
Eight patients did not have a suitable zone 0 for implantation of a thoracic endograft. In these patients, the ascending aorta was replaced to create a Dacron zone 0 landing zone. Therefore, these patients required full cardiopulmonary bypass and a short period of circulatory arrest for simple open distal anastomosis, using both antegrade and/or retrograde cerebral perfusion strategies. Mean CPB time was 259±54 minutes, mean cross-clamp time was 121±63 minutes. Mean circulatory arrest time for distal anastomosis was 19±10 minutes, with an additional 45±13 minutes of selective antegrade cerebral perfusion for arch debranching (Table 3).
30 day/in-hospital mortality was 3/36 (8%) for the entire group (Table 4). Mortality was 11% (n=3) for the Type I group, and zero for the Type II group. Causes of death were all secondary to neurological complications: two from spinal ischemia resulting in permanent paraplegia, and one from complications of stroke. While there have been no in-hospital deaths to date in patients with Type II hybrid arch repairs, this and all other immediate postoperative outcomes showed no significant difference between the two hybrid arch Types (Table 4). 2/36 patients (6%) experienced permanent paraplegia/paraparesis postoperatively, with an additional 5/36 (14%) experiencing mild fully reversed transient neurological events. Importantly, both cases of permanent paraplegia occurred during the first year of adopting this surgical modality.
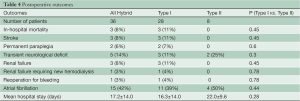
Full table
Late Type II endoleak (branch vessel) occurred in 1 patient, at 3 years post-operatively. Of note, this patient received a Type I hybrid arch repair for proximal degeneration of a prior TEVAR repair (with additional distal TEVAR for distal Type I endoleak in the same operation), and did not receive full relining of the prior endograft repair. This patient did not receive re-intervention and died at 6 years post-operation.
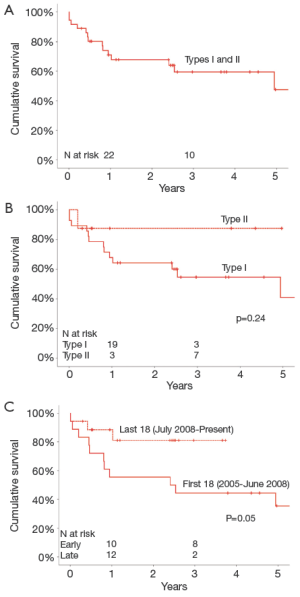
Mean follow-up was 30±21 months. Late freedom from all-cause mortality in the entire cohort was 71%, 60% and 48% at 1, 3 and 5 years (number at risk =22, 10, 4). All but one of the 16 late patient deaths occurred in the Type 1 hybrid group (Figure 2A). Cumulative survival at 1 and 3 years for Type II hybrids was 87%, better than Type I hybrids (68% and 55%, P=0.24) (Figure 2B). We compared outcomes during our early experience with the hybrid arch technique (2005 to June 2008) versus more recent experience (July 2008 to 2012). The early group patients had lower 1 and 3 year survival than the more recent group of patients (56% versus 88% at 1 year, 44% versus 81% at 3 years, P=0.05) (Figure 2C).
The aortic reoperation rate was 1/36 (2.8%). 1 patient required a reintervention for retrograde Type A dissection at 155 days after his Type I hybrid repair - this repair was tolerated with no complications. Importantly, this single retrograde Type A dissection was with a 42 mm barbed endograft for an ascending aortic diameter >38 mm, which in our present algorithm is contraindicated unless a Type II repair is performed.
Discussion
Hybrid approaches for the treatment of aortic arch aneurysmal pathology are being performed with increasing frequency. This is a reflection of the increasing comfort level of the cardiothoracic surgeons with endovascular technology, and the improving technology of endovascular platforms to successfully land stent grafts in the proximal thoracic aorta. As the technology advances, it will be critical for cardiothoracic surgeons to be trained in advancing the field of hybrid aortic arch and ascending aorta operations. As the patient population with thoracic aortic disease seeking intervention get older and older, embracing and honing these skills will be crucial.
In addition to the associated operative mortality of complex operations such as aortic arch hybrid procedures, the Achilles heel of this intervention remains neurologic complications. Several groups have shown that arch hybrid procedures can be performed with acceptable mortality, with very minimal postoperative and long-term endoleak rates (9-15). Neurologic complications, including stroke and spinal cord ischemia, remain significant causes of morbidity and associated mortality. In a systematic review of hybrid arch operations of 1,886 patients, pooled mortality was 10.8%, with 15.1% mortality in diseases that extended to the ascending aorta (16). Pooled stroke risk was 7%, and pooled spinal cord ischemia rate of 7%, with no difference seen with respect to the extent of proximal ascending aortic involvement. In our series of 36 patients, mortality was 8% (n=3), with all deaths seen in the Type I hybrid arch group. Eight patients undergoing Type II hybrid repair had zero mortality. Stroke rate was 8%, with paraplegia rate of 5.5%. All 3 deaths in the cohort had associated neurologic complications (2 with paraplegia, 1 with stroke), highlighting the significant morbidity and mortality associated with this devastating complication in aortic arch surgery. Of note, 42% of the patients in our study had a preoperative history of stroke. 29 out of the 36 patients had atherosclerotic aneurysms, with 52% of the patients having high grade atheroma or mobile atheroma. Atherosclerotic disease is a predictor of perioperative stroke and the large cohort of patients with prior stroke and high grade atheroma (grade ≥3) attests to the comorbid status in the arch hybrid group (12,13). At our institution, the arch hybrid group had a higher rate of high-grade atheroma or mobile atheroma compared to the total arch group (48% versus 6%). Several groups report stroke rates up to 12% in patients undergoing total arch repair of atherosclerotic aneurysms (3,11-14). Our study, in addition to others, suggests that hybrid arch approach to atherosclerotic aneurysmal arch disease can be safely adopted with at least equivocal neurologic outcomes to conventional open repair.
A complication unique to the hybrid aortic arch cohort of patients, unlike the conventional open group, is the occurrence of endoleaks. Literature reporting endoleak rates with hybrid arch repair is not well defined, with long-term follow-up data being virtually absent. Endoleak rates have ranged from 0% to 15% (9-11,15-18). Similar to TEVAR, hybrid arch operations associated with Types I and III endoleak are associated with greater morbidity than Type II endoleak. In a report by Kotelis et al. (13), patients undergoing hybrid arch repair with zone 0 proximal landing had lower endoleak rates than zone 1 landing. At our institution, we do not perform any zone 1 proximal landing. All arch aneurysms are treated for zone 0 landing. In this study, intraoperative endoleak rate was 5/36 (13.8%). All these patients underwent further endovascular reintervention, with a resulting postoperative endoleak rate of 0%. On late follow-up, one patient developed a retrograde Type A dissection from the proximal landing zone site. This case was performed during our fourth year of arch hybrid operations, with the use of a TX2 device (Cook Medical, Bloomington IN). After this experience, we stopped the use of TX2 stent graft device for arch hybrid operations. We have adopted primarily the use of the Gore TAG device (W.L. Gore & Associates, Flagstaff, AZ), and at times the Valiant device (Medtronic, Minneapolis, MN). Since this switch, we have not had a retrograde Type A dissection, and we have had zero distal Type B dissections in the arch aneurysm cases. In other series, in patients with endoleak, the aortic reoperation rate is increased. Our aortic reoperation rate is 1/36 (2.7%) (retrograde Type A dissection with use of TX2 device proximally) over the median midterm follow up of 30 months.
Eight patients in this study underwent Type II arch hybrid repair. Due to the required ascending aortic reconstruction, all these patients had a period of circulatory arrest. Even though these patients had a more complex operation, postoperative and midterm outcomes in these patients have been excellent. Postoperative mortality, stroke, paraplegia, and endoleak rates were zero. In fact, in our experience midterm outcomes of Type II repair have been improved compared to Type I repair outcomes, although not significantly. One patient died at follow-up. As these cases were performed more recently, this may be a reflection of the increasing experience we gained with the hybrid arch platform. In line with this idea, since our first year of hybrid arch operations, the paraplegia and aortic reoperation rates have remained at zero in the Type I arch hybrid group as well, with a single postoperative mortality since then. When we compared overall survival in the early cohort (July 2005 to June 2008) to the late cohort (July 2008 to June 2012), midterm outcomes were superior in the late cohort, even though this group involved more patients requiring a Type II arch hybrid repair. Our experience attests to the learning curve involved with the arch hybrid operation, and that with experience, even the more complex (Type II versus Type I) arch hybrid procedures can be performed with low mortality and morbidity, and good midterm outcomes.
Acknowledgements
Disclosure: The authors declare no conflict of interest.
References
- Svensson LG, Kim KH, Blackstone EH, et al. Elephant trunk procedure: newer indications and uses. Ann Thorac Surg 2004;78:109-16; discussion 109-16. [PubMed]
- Svensson LG, Crawford ES, Hess KR, et al. Deep hypothermia with circulatory arrest. Determinants of stroke and early mortality in 656 patients. J Thorac Cardiovasc Surg 1993;106:19-28; discussion 28-31. [PubMed]
- Sundt TM 3rd, Orszulak TA, Cook DJ, et al. Improving results of open arch replacement. Ann Thorac Surg 2008;86:787-96; discussion 787-96. [PubMed]
- Zeeshan A, Woo EY, Bavaria JE, et al. Thoracic endovascular aortic repair for acute complicated type B aortic dissection: superiority relative to conventional open surgical and medical therapy. J Thorac Cardiovasc Surg 2010;140:S109-15; discussion S142-S146.
- Frigiola A, Ranucci M, Carlucci C, et al. The Ross procedure in adults: long-term follow-up and echocardiographic changes leading to pulmonary autograft reoperation. Ann Thorac Surg 2008;86:482-9. [PubMed]
- Neschis DG, Moainie S, Flinn WR, et al. Endograft repair of traumatic aortic injury-a technique in evolution: a single institution’s experience. Ann Surg 2009;250:377-82. [PubMed]
- Szeto WY, Moser WG, Desai ND, et al. Transapical deployment of endovascular thoracic aortic stent graft for an ascending aortic pseudoaneurysm. Ann Thorac Surg 2010;89:616-8. [PubMed]
- Metcalfe MJ, Karthikesalingam A, Black SA, et al. The first endovascular repair of an acute type A dissection using an endograft designed for the ascending aorta. J Vasc Surg 2012;55:220-2. [PubMed]
- Bavaria J, Milewski RK, Baker J, et al. Classic hybrid evolving approach to distal arch aneurysms: toward the zone zero solution. J Thorac Cardiovasc Surg 2010;140:S77-80; discussion S86-91.
- Weigang E, Parker J, Czerny M, et al. Endovascular aortic arch repair after aortic arch de-branching. Ann Thorac Surg 2009;87:603-7. [PubMed]
- Szeto WY, Bavaria JE, Bowen FW, et al. The hybrid total arch repair: brachiocephalic bypass and concomitant endovascular aortic arch stent graft placement. J Card Surg 2007;22:97-102; discussion 103-4. [PubMed]
- Ergin MA, Galla J, Lansman SL, et al. Hypothermic circulatory arrest in operations on the thoracic aorta: determinants of stroke and early mortality. J Thorac Cardiovasc Surg 1994;107:788-99. [PubMed]
- Gutsche JT, Cheung AT, McGarvey ML, et al. Risk factors for perioperative stroke after thoracic endovascular aortic repair. Ann Thorac Surg 2007;84:1195-200; discussion 1200. [PubMed]
- Estrera AL, Miller CC 3rd, Porat EE, et al. Staged repair of extensive aortic aneurysms. Ann Thorac Surg 2002;74:S1803-5; discussion S1825-32.
- Kotelis D, Geisbüsch P, Attigah N, et al. Total vs hemi-aortic arch transposition for hybrid aortic arch repair. J Vasc Surg 2011;54:1182-6.e2.
- Cao P, De Rango P, Czerny M, et al. Systematic review of clinical outcomes in hybrid procedures for aortic arch dissections and other arch diseases. J Thorac Cardiovasc Surg 2012;144:1286-300, 1300.e1-2.
- Lee CW, Beaver TM, Klodell CT, et al. Arch debranching versus elephant trunk procedures for hybrid repair of thoracic aortic pathologies. Ann Thorac Surg 2011;91:465-71. [PubMed]
- Hughes GC, Daneshmand MA, Balsara KR, et al. “Hybrid” repair of aneurysms of the transverse aortic arch: midterm results. Ann Thorac Surg 2009;88:1882-7; discussion 1887-8.