Comparison of the stand-alone Cox-Maze IV procedure to the concomitant Cox-Maze IV and mitral valve procedure for atrial fibrillation
Introduction
The Cox Maze (CM) procedure was originally developed by Dr. James Cox in 1987. After several revisions to its original lesion set, the Cox-Maze III (CMIII) became the gold standard in the surgical treatment of atrial fibrillation (AF). Initially, it was commonly performed only as a stand-alone procedure because of its technical complexity and long cross clamp time (1,2). As experience grew with the procedure, its use was more broadly applied and began to be performed in patients with concomitant mitral disease and other cardiac pathology. The overall freedom from symptomatic AF in patients who underwent CMIII has been reported to exceed 90% in series with more than five years of follow-up (1,2).
To simplify the CMIII, different ablation technologies were studied in an attempt to reproduce the transmural lesions of the “cut-and-sew” method of the CMIII. This resulted in the development of the Cox-Maze IV (CMIV) in 2002. The CMIV utilized a bipolar radiofrequency energy clamp to achieve continuous transmural ablations and replaced the majority of the “cut-and-sew” lesions of the CMIII (3). This method has proved to be as effective as the CMIII in long-term follow-up while decreasing cross clamp times. The CMIV has become the current standard procedure for the surgical treatment of AF at our institution (4).
AF is present in up to a third of patients undergoing mitral valve (MV) procedures (5). While common, the mechanistic relationship between mitral regurgitation and AF is poorly understood, compared to patients with lone AF. Moreover, many of the early case series, including our own, concentrated on results in patients with lone AF (4). With the advent of the CMIV and its decreased operative times, over half of patients currently receiving a CMIV procedure are undergoing concomitant MV surgery, highlighting the significant role of AF procedures (5). Given that the CMIV operation is performed in a concomitant fashion the majority of the time, it is critical to evaluate outcomes in this population. The purpose of this study was to compare outcomes of the stand-alone CMIV procedure to the CMIV procedure with concomitant mitral procedure and identify risk factors for recurrence of AF.
Methods
This study was approved by the Washington University School of Medicine Institutional Review Board. Informed consent and permission for release of information was obtained from each participant. All data were entered prospectively into a longitudinal database maintained at our institution.
Patients
A total of 335 patients, from January 2002 to December of 2012, who received a CMIV procedure for AF as a stand-alone CMIV (n=151) or concomitantly with a MV procedure (n=184) were reviewed. The CMIV procedure has previously been described (3). All patients received pulmonary vein isolation (PVI), confirmed by exit block from each pulmonary vein, unless patients had a left atrial (LA) thrombus which precluded cardioversion or cardioversion was unsuccessful. The lesion set was similar among all patients except for the addition in 2005 of a superior LA lesion which connected the right and left superior pulmonary veins. This lesion effectively electrically isolated the entire posterior left atrium and created a “box-lesion” set. A small set of patients (n=19) underwent a LA lesion set only.
Patients were discharged on class I or III antiarrhythmic drugs and warfarin, unless contraindicated; antiarrhythmic agents were discontinued two months postoperatively if patients were in normal sinus rhythm. β-blockers or calcium channel blockers were not considered as antiarrhythmic drugs.
Study design
Demographics and perioperative variables including complications were compared between stand-alone CMIV and concomitant groups. Early atrial tachyarrhythmias (ATAs) were defined as any ATA documented within 30 days. Patients were evaluated starting at three months for recurrent ATAs. Electrocardiographic data were obtained at 3, 6, 12, 24, and 48 months. Initially, this consisted of either ECG or 24-hour Holter monitoring. Starting in the 2008, 24-hour continuous cardiac monitoring was routinely obtained in all patients (6).
Late recurrence was defined as any episode of AF, atrial flutter or atrial tachycardia that lasted greater than 30 seconds (6). Patients that required an interventional procedure were deemed permanent failures.
Statistical analysis
Continuous variables are expressed as mean ± standard deviation (SD) or as median with range. Categorical variables are expressed as frequencies and percentages with outcomes compared using the χ2 or the Fisher exact test. Continuous outcomes were compared using the t-test for means of normally distributed continuous variables and the Mann-Whitney U nonparametric test for skewed distributions.
Twenty-four preoperative and perioperative variables were evaluated by univariate analysis to identify predictors of AF recurrence at one year. Variables used in univariate analyses included the presence of a concomitant procedure in addition to those listed in Tables 1 and 2, with the exception of mortality, intensive care unit (ICU) and hospital length of stay (LOS). Significant covariates on univariate analysis (P≤0.10) or covariates deemed clinically relevant based on experience were entered into a multivariate binary logistic regression analysis. All data analyses were performed using SYSTAT 13 software (Systat Software, Inc., Chicago, IL).
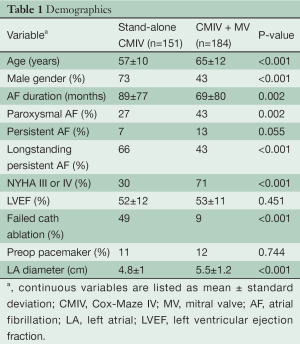
Full table
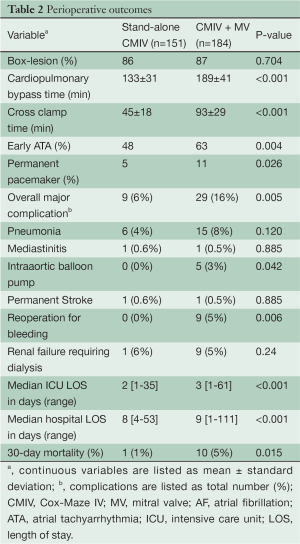
Full table
Results
A total of 151 patients (45%) underwent stand-alone CMIV procedure and 184 (55%) underwent the CMIV with a concomitant MV operation. MV operations included patients receiving a MV repair (111 patients, 60%) or MV replacement (73 patients, 40%). Twenty-nine patients receiving a mitral operation also underwent a tricuspid valve procedure (Table 3).
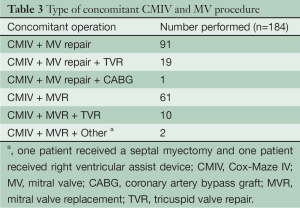
Full table
Follow-up was available on 72% of patients at one year and 55% of these at two years. Twenty-four hours of continuous monitoring was performed in 75% of patients who had follow-up after 2008 (6). Mean follow-up time was 2.7±2.4 years.
Demographics
Demographics showed significant preoperative differences in terms of age, gender, AF duration, type of AF, NYHA classification, presence of failed catheter ablation and LA diameter between the two groups (Table 1). Notably, patients with MV disease had a longer documented duration of AF by 20 months and a higher incidence of longstanding persistent AF. Ejection fraction was similar between concomitant and stand-alone CMIV groups (53% vs. 52%, P=0.451) despite a greater percentage of patients with NYHA class III or IV symptoms in the concomitant group (71% vs. 30%, P
Perioperative outcomes
Perioperative differences including complications and 30-day mortality can be seen in Table 2. As expected, cardiopulmonary bypass (CPB) time was significantly longer in the concomitant group (189 vs. 133 min, Pvs. 45 min, Pvs. 6%, P=0.005), with significant differences between the need for an IABP (3% vs. 0%, P=0.042) and reoperation for bleeding (5% vs. 0%, P=0.006). Early post-operative ATAs were more frequent in the concomitant group (63% vs. 48%, P=0.004).
Median ICU LOS was decreased in the stand-alone CMIV group (2% vs. 3%, Pvs. 9%, Pvs. 5%, P
Efficacy
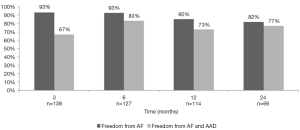
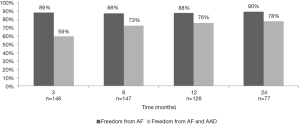
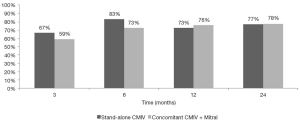
Freedom from ATA after a stand-alone CMIV procedure was 93%, 93%, 85%, and 82% at 3, 6, 12, and 24 months respectively. Freedom from ATA and antiarrhythmic drugs was lower at each time point, but the differences decreased with increasing follow-up (Figure 1). When performing a concomitant CMIV in addition to a MV procedure, freedom from ATA was 89%, 88%, 88%, and 90% at 3, 6, 12, and 24 months respectively (Figure 2). Comparisons between the two groups showed no significant differences after six months (Figure 3).
Multivariate analysis
In all patients, predictors of recurrence were having a preoperative pacemaker (P
Discussion
In this study, perioperative outcomes and late efficacy were compared between patients having a stand-alone CMIV and those having concomitant mitral surgery. Previous studies from our institution and others usually have compared the stand-alone Cox-Maze procedure to a combined cohort of patients receiving a Cox-Maze procedure in addition to multiple concomitant procedures, including coronary artery bypass grafting, aortic valve replacement, and MV repair (2,7). Because the diseases prompting these concomitant operations are the result of multiple heterogenous pathologies, it is difficult to interpret the results from a single “concomitant” cohort. It was for this reason that this study was designed to compare outcomes in patients with lone AF to those with AF and concomitant mitral disease. This comparison is important for two reasons. First of all, AF ablation is performed much more commonly in the setting of concomitant mitral disease than for lone AF. In a recent study, five times as many surgical ablations were performed in the US in patients undergoing mitral surgery than for lone AF (5). Second, patients with MV disease often have enlarged LA from either chronic volume or pressure overload. This may result in different mechanisms of AF in this population, which could impair procedural success. Studies have shown that LA diameter is a risk factor for late recurrence of AF after a Cox-Maze procedure (8).
In our study, the efficacy of the CMIV for both stand-alone AF and when combined with a MV procedure was similar with respect to freedom from AF and antiarrhythmic agents at both one and two years. This is in agreement with our previous data showing comparable efficacy at one year using either the CMIII or the CMIV procedure, and shows that these good results were maintained at two years (2,9).
As expected, the two groups had different demographics. Patients who received stand-alone CMIV procedure tended to be predominantly male, younger and had a higher incidence of longer standing persistent AF. Moreover, half of the patients had failed a previous catheter ablation. The concomitant group on the other hand had more patients with NYHA class III or IV symptoms and significantly larger LAs. Despite these differences, the efficacy of the CMIV was similar in both groups, highlighting the efficacy of the CMIV procedure for the treatment of AF in both populations.
Not surprisingly, there was a significant difference in the perioperative morbidity, mortality and 30-day mortality between the groups. In a previous report from our institution, 30-day mortality was not significant (9). However, the total number of patients in this study was increased by 57% compared to our previous publication.
Major complication rates were also significantly increased in the concomitant group, primarily because of the need for reoperation for bleeding and an IABP. This is consistent with the fact that patients undergoing concomitant MV surgery were older, sicker and had longer cross clamp and CPB times. This increase in overall complication rate likely accounts for the one day increase in both ICU and hospital LOS.
The multivariate analysis of risk factors for recurrent ATA in this large cohort of over 350 patients was informative. In agreement with prior studies from our group, the failure to perform a box-lesion around the pulmonary veins and posterior left atrium was a predictor of recurrence. The increased risk of recurrence seen with a preoperative pacemaker is a new observation and is likely due to the improved power of this study in detecting smaller differences in outcomes. Since the majority of pacemakers were implanted for sick sinus syndrome, this finding suggests that patients with intrinsic sinus node dysfunction may have higher recurrence rates, possibly due to the known increased incidence of premature atrial contractions in these patients which can act as a trigger for recurrent AF.
The presence of early postoperative ATA as a predictor of late recurrence was in agreement with previous reports in the literature (8,10). This finding suggests that part of the substrate for early and late ATA may be similar. However this finding needs to be put into perspective. In patients undergoing concomitant MV and CMIV procedures, early ATA were present in 63% of patient but at two years only 10% of patients had recurrent ATA. Thus, it is important to remember that the majority of patients with early ATA remain free of ATA at two years.
Surprisingly, LA size was not a predictor in this multivariate analysis in contradiction to other studies which have shown this to be an important variable for failure (10,11). This may have been due to the introduction of the box-lesion set. In these patients, isolation of the entire posterior LA may effectively decrease LA size from an electrophysiological standpoint and reduce the substrate available for maintaining AF, thus negating the effect of an enlarged left atrium.
This study has several limitations. While the majority of our patients did have 24-hours of continuous cardiac monitoring after six months, there were still some who only had ECGs leading to a possible underestimation in the incidence of recurrent AF. In addition, this study was a retrospective review which introduces inherent selection biases, that could potentially be avoided if performed in a randomized fashion. However, all data were collected prospectively and this was a series of consecutive patients. While this series did show that efficacy was maintained at two years, it did not determine whether failures were due to an inability to properly complete the lesion set or because the underlying mechanism of AF in these patients was not able to be eliminated with a Cox-Maze procedure. It is our impression that failures occur because of advanced atrial remodeling and subsequent substrate modification such as atrial fibrosis. In this setting, reentry can be sustained in very small areas of atria, making it virtually impossible to eliminate AF with a Cox-Maze procedure (12). Finally, the AF burden was not quantitated in this study. Future studies using implantable loop recorders (ILRs) are worth considering to provide this information.
In conclusion, patients receiving a CMIV procedure have high rates of freedom from AF at one and two years regardless of the addition of a MV procedure. Perioperative complications were increased in patient undergoing concomitant MV surgery resulting in a longer hospital stay and higher 30-day mortality. Further advances in surgical techniques such as minimally invasive approaches may help to offset these differences.
Acknowledgements
Support in part by National Institutes of Health grants R01 HL032257 and T32 HL007776. RJD receives research grants and educational funding from AtriCure, Estech, and Edwards.
Disclosure: RJD is a consultant for AtriCure and Medtronic. Other co-authors declare no conflict of interest.
References
- Cox JL, Ad N, Palazzo T, et al. The Maze-III procedure combined with valve surgery. Semin Thorac Cardiovasc Surg 2000;12:53-5. [PubMed]
- Prasad SM, Maniar HS, Camillo CJ, et al. The Cox maze III procedure for atrial fibrillation: long-term efficacy in patients undergoing lone versus concomitant procedures. J Thorac Cardiovasc Surg 2003;126:1822-8. [PubMed]
- Gaynor SL, Diodato MD, Prasad SM, et al. A prospective, single-center clinical trial of a modified Cox maze procedure with bipolar radiofrequency ablation. J Thorac Cardiovasc Surg 2004;128:535-42. [PubMed]
- Weimar T, Schena S, Bailey MS, et al. The cox-maze procedure for lone atrial fibrillation: a single-center experience over 2 decades. Circ Arrhythm Electrophysiol 2012;5:8-14. [PubMed]
- Gammie JS, Haddad M, Milford-Beland S, et al. Atrial fibrillation correction surgery: lessons from the Society of Thoracic Surgeons National Cardiac Database. Ann Thorac Surg 2008;85:909-14. [PubMed]
- European Heart Rhythm Association (EHRA), European Cardiac Arrhythmia Society (ECAS), American College of Cardiology (ACC), et al. HRS/EHRA/ECAS expert Consensus Statement on catheter and surgical ablation of atrial fibrillation: recommendations for personnel, policy, procedures and follow-up. A report of the Heart Rhythm Society (HRS) Task Force on catheter and surgical ablation of atrial fibrillation. Heart Rhythm 2007;4:816-61. [PubMed]
- Stulak JM, Sundt TM 3rd, Dearani JA, et al. Ten-year experience with the Cox-maze procedure for atrial fibrillation: how do we define success? Ann Thorac Surg 2007;83:1319-24. [PubMed]
- Damiano RJ Jr, Schwartz FH, Bailey MS, et al. The Cox maze IV procedure: predictors of late recurrence. J Thorac Cardiovasc Surg 2011;141:113-21. [PubMed]
- Saint LL, Bailey MS, Prasad S, et al. Cox-Maze IV results for patients with lone atrial fibrillation versus concomitant mitral disease. Ann Thorac Surg 2012;93:789-94. [PubMed]
- Ad N, Henry L, Holmes SD, et al. The association between early atrial arrhythmia and long-term return to sinus rhythm for patients following the Cox maze procedure for atrial fibrillation. Eur J Cardiothorac Surg 2013;44:295-300. [PubMed]
- Kim KC, Cho KR, Kim YJ, et al. Long-term results of the Cox-Maze III procedure for persistent atrial fibrillation associated with rheumatic mitral valve disease: 10-year experience. Eur J Cardiothorac Surg 2007;31:261-6. [PubMed]
- Byrd GD, Prasad SM, Ripplinger CM, et al. Importance of geometry and refractory period in sustaining atrial fibrillation: testing the critical mass hypothesis. Circulation 2005;112:I7-13. [PubMed]





