Clinical psychological and neuropsychological issues with left ventricular assist devices (LVADs)
Introduction
Heart transplantation is the treatment of choice for many patients with end-stage heart failure (HF). However the limited availability of organs has sparked interest in developing alternative approaches to myocardial replacement. Recently, a second generation of continuous-blood-flow pumps (CBFP) have been introduced, furthering ventricular assist devices (VADs) as an alternative form of life-saving therapy. Initially designed as a temporary support to bridge patients to heart transplant, these devices are increasingly being used as lifetime support or destination therapy (1).
VADs offer life-saving therapy and improved quality of life (QOL) for patients with end-stage HF. However, even with this therapy, patients are not free of complications as the prolongation of life is not always synonymous with improved quality despite the good intentions of medical practitioners. Patients are in fact continuously exposed to neurological events, infections and right HF, which could negatively influence their neuropsychological and QOL status (2-4). Furthermore, the mortality following left ventricular assist device (LVAD) implantation remains significant, at approximately 30% by two years (5).
Currently, the literature on the cognitive and psychological status of LVAD patients is still in its infancy and has two main limitations: (I) cognitive and psychological aspects are studied separately without considering their mutual influences; and (II) studies tended to focus only on the first year after LVAD implantation without taking into account the pre-implantation status and its evolution over time. What is actually needed is the continuous monitoring of patients’ physical and psychological profiles to allow intervention in case of negative outcomes.
In regards to cognitive profile, it has been found that patients show slight improvements within the first 24 months after CBFS (6,7), but a baseline neurocognitive assessment before implantation and the impact of these dysfunctions on daily life have not been considered. Assessments of cognitive functions pre- and post-LVAD implantation are needed for several reasons. Firstly, low cardiac output associated with HF often leads to end-organ damage, including brain injury; HF patients can have cognitive deficits ranging from mild to severe (8). The presence of cognitive deficits could compromise patients’ capacity to comprehend and participate in the decision-making process before surgery. Furthermore, cognitive deficits can influence the ability to understand and adhere to post-LVAD treatment protocols. By affecting the basic activities of daily living, cognitive disorders also impact on caregivers’ QOL.
Data from the current literature reported a better QOL at one month to one year post-implantation in LVAD patients (9-11), but a worse health-related QOL (HRQOL) when compared to HTx patients (12,13). QOL is also related to psychological factors (i.e., anxiety and depression) which are good predictors of patient satisfaction and wellbeing (9). In addition, LVAD patients tend to face many emotional problems, as they have to accept a life that is far from normal and modify their lifestyle. That is, they must co-exist with a mechanical device attached to their body, taking care of and treating it like a real part of their body. Patients have been shown to experience fatigue, anxiety, depression and sleep disturbances up to six months after implantation (14,15).
LVADs also have serious negative effects for caregivers, who must manage many of the patient’s daily problems. Caregivers are at higher risk of psychological distress, as their responsibility to patients can impose limitations on their own lives including their relationships and careers (16-19).
Patients with LVADs represent a new population which must be more extensively studied to help clinicians better understand their needs and improve their QOL. The aims of the present research are therefore two-fold: (I) to develop clinical guidelines and pathways which support the implementation of best practice in the assessment of LVAD patients, by considering cognitive, psychological and emotional aspects as well as caregiver status; (II) to study the evolution of these aspects during both the preoperative and postoperative LVAD support periods, and better monitor for adverse changes. In fact, although LVADs offer excellent survival and QOL, many complications can occur and this requires extensive medical care and follow-up. LVADs will play an increasing role in the near future. However, there is still much to learn in regards to patient care, especially concerning device-specific complications.
Materials and methods
The ethics committee of the Hospital of Padova approved the study. All clinical investigations have been conducted consistent with the declaration of Helsinki. A longitudinal, repeated-measures, case-control design was used. A sample of 19 patients (mean age: 60.4±10 years; years of education: 10.9±3.6; 17 males) with end-stage cardiac disease who underwent routine psychological and neuropsychological evaluation prior to LVAD implantation was studied at Time 0 (T0). The demographic and clinical characteristics are illustrated in Table 1.

Full table
A subsample of 10 patients was reassessed twice, after the LVAD implantation, at Time 1 (T1) (2.5±2.4 months), and at Time 2 (T2) (15.6±5.8 months) (see Table 2).

Full table
The devices currently used in our hospital are “Jarvik 2000” as destination therapy and “HeartWare” as bridge-to-transplantation, both CBFP.
Psychological and QOL measures
Prior to LVAD implantation, patients underwent a psychological assessment based on a 45-minute semi-structured interview and two questionnaires: the Minnesota multiphasic personality inventory (MMPI-2) (20) and the 36 item short form health survey (SF-36) (21). The MMPI-2 is an effective and reliable test which evaluates the most prominent features of an individual’s personality. It is a self-administered questionnaire to which the subject must answer “true” or “false” to a series of 567 statements about his/her personal experiences, physical conditions, habits, and attitude. It constructs an exhaustive clinical picture of the patient by means of a series of numerical variables, and has a high discriminative power to discern between normal and pathological traits of personality. The test involves three scales of validity (L, lie; F, infrequency; K, correction), 10 base clinical scales (Hs, hypochondriasis; D, depression; Hy, hysteria; Pd, psychopathic deviate; Mf, masculinity–femininity; Pa, paranoia; Pt, psychasthenia; Sc, schizophrenia; Ma, hypomania; Si, social introversion), and 15 clinical content scales (ANX, anxiety; FRS, fears; OBS, obsessiveness; DEP, depression; HEA, health concerns; BIZ, bizarre mentation; ANG, anger; CYN, cynicism; ASP, antisocial practices; TPA, type A; LSE, low self-esteem; SOD, social discomfort; FAM, family problems; WRK, work interference; TRT, negative treatment indicators). For psychological diagnostic purposes, the entire profile is determined from both the 10 base scales and the 15 subject scales in order to achieve maximum discriminating power. The scales of the test are standardized according to a normal distribution (T scores); scores above the 65th centile are considered clinically significant.
QOL was assessed by the SF-36, which evaluates some prominent conceptions about health, independent of age, health status, and treatment. The SF-36 has one multi-item scale that assesses eight health domains: physical functioning (PF); physical role (PR); bodily pain (BP); general health (GH); vitality (VT); social functioning (SF); emotional role (ER); mental health (MH). The SF-36 test has an acceptable internal consistency and retest reliability. In our study we used the well-validated Italian version of SF-36 (21). SF-36 scores were converted to a scale of 0 to 100 (a higher score indicates a better QOL).
After LVAD implantation, only SF-36 was re-administered. In addition, the Symptom Checklist 90 (SCL-90) (22), which reflects psychological symptom patterns of psychiatric and medical patients, was introduced. The subscales for this instrument are somatization (SOM), obsessiveness-compulsiveness (OC), interpersonal sensitivity (IS), Depression (DEP), ANX, hostility (HOS), phobic anxiety (PHOB), paranoid ideation (PAR), psychoticism (PSY) and sleep disturbances (SLEEP). There is a global index of distress, the Global Severity Index (GSI), which is a general indicator of the current level of a patient’s psychological distress.
The SF-36 was also administered to caregivers at T0, T1 and T2, to quantify their QOL.
Neuropsychological measures
Prior to LVAD implantation, patients underwent routine neuropsychological assessment by the Mini Mental State Examination (MMSE) (23) and by a well-validated battery for the Italian population, the Esame Neuropsicologico Breve 2 (ENB2, Brief Neuropsychological Examination 2) (24). Each patient was evaluated individually by experienced neuropsychologists and the assessment lasted one hour.
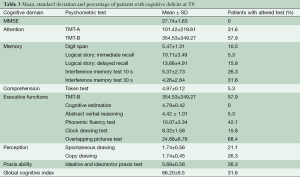
The MMSE consists of a brief 30-point questionnaire used to screen cognitive decline (cut-off score: 24). The ENB2 battery investigates different cognitive domains and includes 16 subtests: digit span, immediate and delayed recall prose memory, interference memory (10 and 30 seconds), Trail Making Test parts A and B, token test (five items), word phonemic fluency, abstraction, cognitive estimation, overlapping figure, spontaneous drawing, copy drawing, clock drawing and ideative and ideomotor praxis tests. An ENB2 total score was calculated to obtain a general measure of the cognitive status. The tests were chosen to cover five cognitive domains: attention, memory, executive functions, and perceptive and praxis abilities. The cognitive domain of attention included the Trail Making Test A and the Trail Making Test B (TMTB); the domain of memory included digit span, logical story and interference memory tests; the cognitive domain of executive function included Trail Making Test B, cognitive estimation, abstract reasoning, phonemic fluency, clock drawing, and overlapping pictures tests. The domain of perception included Spontaneous drawing and copy drawing tests. One test accounted for more than one domain (Table 3): TMTB is a well-known instrument for describing the attentive function but at the same time it evaluates switching ability and working memory (i.e., executive functions); thus, it requires the involvement of executive functions (25).
Full table
After LVAD implantation, both the MMSE and ENB2 were re-administered (see Figure 1).
Statistical analysis
Data were analyzed using descriptive statistics, Spearman correlation, analysis of variance with repeated measures and Wilcoxon Test. The level of significance was set at P≤0.05.
Psychological and QOL at T0
None of the 19 patients had any abnormal scores at MMP2, showing that that patients did not show any psychopathological depression (scale D) and anxiety (scales Pt and A).
QOL at T0
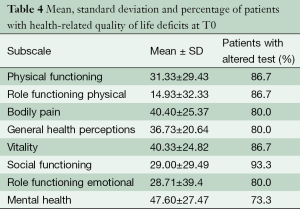
Means, SDs and percentages of patients with altered SF-36 subtests are summarized in Table 4. Patients showed a global reduction in QOL in all the SF-36 scales, reflecting the severe impact of congestive HF on daily life. This is likely due to the severity of the illness, with symptoms such as fatigue and breathlessness, which may limit physical and SF. This interpretation is supported by findings of Ekman and co-workers (26) and Komaroff and co-workers (27) who presented similarly low SF-36 scores to those in this study.
Full table
No correlations were found between cognitive and QOL measures coherently with other studies (28,29).
QOL at T1 and T2
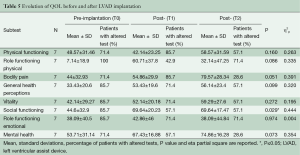
Means, SDs and percentages of patients with altered SF-36 subtests are summarized in Table 5. No significant improvements were found. However, although the P value was not significant, the high value of partial eta square could suggest that this lack of statistical significance could be inherently due to the study’s small sample size.
Full table
SCL-90 at T1 and T2
Means, SDs and percentages of patients with altered SCL-90 subtests are summarized in Table 6. No significant changes were found.

Full table
Neuropsychological results at T0
Results showed that 94.7% of the patients had a normal level of comprehension, which is consistent with the high reliability of cognitive tests. The mean MMSE score for the total patient sample was 27.7±1.6. None of the patients showed impaired MMSE scores but this is unsurprising, given that the MMSE may detect cognitive impairment less effectively due to the ceiling effect, differences in educational background, and the absence of specific tests of executive functions. In fact, detailed neuropsychological evaluation showed that 26.3% failed five or more tests, indicating broad neuropsychological impairment. In detail, executive functions were impaired in 89.5%, perception in 36.8%, memory in 57.9%, attention in 57.9%, and praxis abilities in 26.3% of all patients (Table 3). Our results confirm data from the literature showing that patients with severe HF suffer from cognitive impairment in one or more cognitive domains, which may compromise adherence to medical treatments (8,29) and result in diminished QOL. Circulatory insufficiency leading to inadequate cerebral perfusion and cerebral hypoxia is one of the most likely etiologies of cognitive deficits among adults with chronic HF (30,31).
Neuropsychological results at T1 and T2
The results of cognitive performance before and after the LVAD are summarized in Table 7. Comparisons of the neuropsychological assessment results at T0, T1 and T2 showed significant improvements over time in the domain of memory [logical story immediate recall: (T0 vs. T2 and T1 vs. T2) F[2,18[ =6.123, P=0.009; η2p=0.405; interference memory 10 s (T1 vs. T2): F[2,18] =3.988, P=0.37, η2p=0.307; interference memory 30 s (T0 vs. T2): F[2,18] =3.915, P=0.039; η2p=0.303]. For the other cognitive domains, patient profiles tended to remain stable over time without significant improvements. Despite the constant risk of neurological events associated with the CFBP, patients’ cognitive functions do not get worse and in fact significant enhancements were observed.
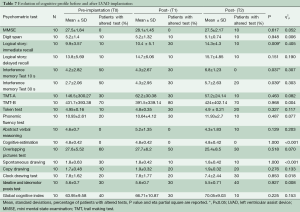
Full table
Correlations
Concerning cognitive measures, no correlations were found between QOL and SCL-90 scores at T1.
Concerning correlations between SF-36 and SCL-90, at T1, PF correlated with anxiety (r=–0.769) and hostility (r=–0.827); BP correlated with anxiety (r=–0.686) and hostility (r=–0.736); VT correlated with OC (r=–0.775), anxiety (r=–0.809) and hostility (r=–0.886); SF correlated with depression (r=–0.788); mental health with hostility (r=–0.866) and anxiety (r=–0.738). At T2, PR correlated with SOM (r=–0.876); physical pain with SOM (r=–0.786), OC (r=–0.798) and depression (r=–784); GH with SOM (r=–0.670), VT with SOM (r=–0.884); ER with SOM (r=–0.701) and psychoticism (r=–0.737); mental health with OC (r=–0.698) and hostility (r=–0.715). At T2, significant correlations between GSI and the subscale of PR (r=–0.717), OC (r=–0.796) and depression (r=–0.778) were found.
Caregivers’ results
Table 8 shows the means, SDs and percentages of caregivers with SF-36 altered subtests. In addition, correlations between patients’ and caregivers’ QOL are performed. A significant correlation was found at T2 between patients’ GH and caregivers’ PR and social activities (r=0.861 and r=0.832 respectively); between patients’ social activities and caregivers’ PR and social activities (r=0.939 and r=0.893 respectively); and between patients’ ER and caregivers’ PR (r=0.839) and social activities (r=0.889).
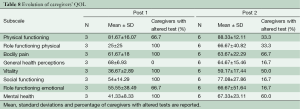
Full table
General discussion
With the reduced availability of donor hearts for patients with severe HF, LVADs have been introduced not only as a bridge-to-transplantation but also as a destination therapy. Continuous-flow pumps appear to have some advantages compared to pulsatile-flow both in survival and complication rates (32). On one hand, such devices are essential for the survival of end-stage HF patients for whom a heart transplant is not readily available. On the other hand, such devices also expose patients to a constant risk of neurological events and death. Thus, it becomes essential to monitor patients’ cognitive and psychological statuses in order to maximize benefits of the therapy and enhance QOL.
Our aim was to therefore holistically evaluate patients, considering cognitive, psychological and emotional status together because of their mutual influence (33). Additionally, we have highlighted the importance of evaluating patients not only post-LVAD implantation, as usually reported in the literature, but also pre-implantation, in order to monitor the neurobehavioral trend of patients during LVAD therapy.
Periodical follow-up should be introduced as it could help identify any initial psychological problems or deficits that could negatively influence patient management. Our preliminary data showed that patients’ cognitive, psychological and QOL status tended to remain stable from pre- to post-LVAD implantation. In addition, significant improvements were found in the domain of memory.
Despite their exposure to continuous flow pumps, patients did not show cognitive decline; on the contrary, they demonstrated an improvement after LVAD which was maintained even after 16 months.
Such results are very important, since recipients must possess the ability to manage their complex treatment regimen. Compliance after LVAD implantation is multifaceted and involves adherence to a prescribed diet and lifestyle, scheduled medical appointments, maintenance of communication, and, ultimately, proper care for batteries. The possible short- and long-term effects of neurocognitive decline on the QOL after cardiac surgery are well-recognized, but it is the subject of an ongoing debate in literature (34).
Significant positive correlations were found between QOL and psychological and emotional aspects. Patients who reported better QOL experienced fewer negative psychological problems. Such data reflect the reciprocal influence of such domains. In fact, Sandau et al. (33) stated that “QOL is a subjective, temporal, multi-dimensional construct and should include domains important to the patients. For LVAD patients, these domains are physical, emotional, cognitive, social, and spiritual with corresponding subdomains”. QOL is a multidimensional concept and includes aspects of physical, mental and SF. Interestingly, the correlations also found between patients’ and caregivers’ QOL agree with the current literature showing that the patients’ psychological well-being is related to support from family (19). Consequently, it is important to provide patients and their families with a program of psychological assistance throughout the LVAD support (before, during and after surgery). At the same time, families should receive clear, simple and concise descriptions of what the implantation entails.
The present study may be limited somewhat by the variable number of patients studied at each time interval due to factors such as death, transplantation, staff availability, or scheduling. Thus, there could be ascertainment bias due to exclusion of sick patients.
In conclusion, we propose the development of a complete psychological assessment that includes emotional, psychopathological, and cognitive aspects. This would improve the assessment of LVAD patients and enable a better QOL for them and their caregivers. In order to prevent adverse outcomes, we suggest that it is essential to keep in constant contact with patients and their families, and guide them through the complex management of treatment. For example, it is vital to facilitate patients in adapting to their device during the early post-implantation period, as fear and anxiety at home is very common at this stage. It is crucial that the patient feels secure with the LVAD, as anxiety regarding management of the machine can precipitate a sense of isolation and the development of psychological disturbances (2). These problems, in addition to other possible neurological complications, may affect treatment success. The results obtained highlight the need for regular, long-term psychological support for this vulnerable patient population, and the importance of further studies exploring possible differences between destination therapy and bridge-to-transplantation patients. We stress the importance of effective multidisciplinary collaboration in order to establish a programme of research and professional education. The clinical practice guideline we propose here could assist the assessment and management of cognitive and neuropsychological alterations in patients with LVAD implantation, while also considering the economic costs of this mechanical implantation.
Acknowledgements
Disclosure: The authors declare no conflict of interest.
References
- Boilson BA, Raichlin E, Park SJ, et al. Device therapy and cardiac transplantation for end-stage heart failure. Curr Probl Cardiol 2010;35:8-64. [PubMed]
- Chapman E, Parameshwar J, Jenkins D, et al. Psychosocial issues for patients with ventricular assist devices: a qualitative pilot study. Am J Crit Care 2007;16:72-81. [PubMed]
- Rizzieri AG, Verheijde JL, Rady MY, et al. Ethical challenges with the left ventricular assist device as a destination therapy. Philos Ethics Humanit Med 2008;3:20. [PubMed]
- Allen JG, Weiss ES, Schaffer JM, et al. Quality of life and functional status in patients surviving 12 months after left ventricular assist device implantation. J Heart Lung Transplant 2010;29:278-85. [PubMed]
- Kirklin JK, Naftel DC, Kormos RL, et al. Fifth INTERMACS annual report: risk factor analysis from more than 6,000 mechanical circulatory support patients. J Heart Lung Transplant 2013;32:141-56. [PubMed]
- Petrucci RJ, Wright S, Naka Y, et al. Neurocognitive assessments in advanced heart failure patients receiving continuous-flow left ventricular assist devices. J Heart Lung Transplant 2009;28:542-9. [PubMed]
- Petrucci RJ, Rogers JG, Blue L, et al. Neurocognitive function in destination therapy patients receiving continuous-flow vs pulsatile-flow left ventricular assist device support. J Heart Lung Transplant 2012;31:27-36. [PubMed]
- Mapelli D, Bardi L, Mojoli M, et al. Neuropsychological profile in a large group of heart transplant candidates. PLoS One 2011;6:e28313. [PubMed]
- Grady KL, Meyer P, Mattea A, et al. Predictors of quality of life at 1 month after implantation of a left ventricular assist device. Am J Crit Care 2002;11:345-52. [PubMed]
- Wray J, Hallas CN, Banner NR. Quality of life and psychological well-being during and after left ventricular assist device support. Clin Transplant 2007;21:622-7. [PubMed]
- Rogers JG, Aaronson KD, Boyle AJ, et al. Continuous flow left ventricular assist device improves functional capacity and quality of life of advanced heart failure patients. J Am Coll Cardiol 2010;55:1826-34. [PubMed]
- Hallas C, Banner NR, Wray J. A qualitative study of the psychological experience of patients during and after mechanical cardiac support. J Cardiovasc Nurs 2009;24:31-9. [PubMed]
- Kugler C, Malehsa D, Tegtbur U, et al. Health-related quality of life and exercise tolerance in recipients of heart transplants and left ventricular assist devices: a prospective, comparative study. J Heart Lung Transplant 2011;30:204-10. [PubMed]
- Casida JM, Parker J. A preliminary investigation of symptom pattern and prevalence before and up to 6 months after implantation of a left ventricular assist device. J Artif Organs 2012;15:211-4. [PubMed]
- Shapiro PA, Levin HR, Oz MC. Left ventricular assist devices. Psychosocial burden and implications for heart transplant programs. Gen Hosp Psychiatry 1996;18:30S-35S. [PubMed]
- Bunzel B, Laederach-Hofmann K, Wieselthaler GM, et al. Posttraumatic stress disorder after implantation of a mechanical assist device followed by heart transplantation: evaluation of patients and partners. Transplant Proc 2005;37:1365-8. [PubMed]
- Bunzel B, Roethy W, Znoj H, et al. Psychological consequences of life-saving cardiac surgery in patients and partners: measurement of emotional stress by the Impact of Event Scale. Stress and Health 2008;24:351-63.
- Kaan A, Young QR, Cockell S, et al. Emotional experiences of caregivers of patients with a ventricular assist device. Prog Transplant 2010;20:142-7. [PubMed]
- Baker K, Flattery M, Salyer J, et al. Caregiving for patients requiring left ventricular assistance device support. Heart Lung 2010;39:196-200. [PubMed]
- Butcher JN, Rouse SV. Clinical personality assessment. Annu Rev Psychol 1996;47:385-401. [PubMed]
- Apolone G, Cifani S, Liberati MC, et al. Questionario sullo stato di salute SF 36. Traduzione e validazione in italiano (Progetto IQOLA) Metodologia e didattica Clinica 1997;5:86-94.
- Derogatis LR. SCL-90-R: administration, scoring and procedures manual II. Towson, MD: clinical psychometric research, 1983.
- Folstein MF, Folstein SE, McHugh PR. “Mini-mental state”. A practical method for grading the cognitive state of patients for the clinician. J Psychiatr Res 1975;12:189-98. [PubMed]
- Mondini S, Mapelli D, Vestri A, et al. Esame neuropsicologico breve 2. Milano: Raffaello Cortina 2011.
- Lezak MD. Neuropsychological assessment.3rd.ed. New York: Oxford University Press, 1995.
- Ekman I, Fagerberg B, Lundman B. Health-related quality of life and sense of coherence among elderly patients with severe chronic heart failure in comparison with healthy controls. Heart Lung 2002;31:94-101. [PubMed]
- Komaroff AL, Fagioli LR, Doolittle TH, et al. Health status in patients with chronic fatigue syndrome and in general population and disease comparison groups. Am J Med 1996;101:281-90. [PubMed]
- Zuccalà G, Cattel C, Manes-Gravina E, et al. Left ventricular dysfunction: a clue to cognitive impairment in older patients with heart failure. J Neurol Neurosurg Psychiatry 1997;63:509-12. [PubMed]
- Bennett SJ, Sauvé MJ. Cognitive deficits in patients with heart failure: a review of the literature. J Cardiovasc Nurs 2003;18:219-42. [PubMed]
- Pullicino PM, Hart J. Cognitive impairment in congestive heart failure?: Embolism vs hypoperfusion. Neurology 2001;57:1945-6. [PubMed]
- Taylor J, Stott DJ. Chronic heart failure and cognitive impairment: co-existence of conditions or true association? Eur J Heart Fail 2002;4:7-9. [PubMed]
- Garbade J, Barten MJ, Bittner HB, et al. Heart transplantation and left ventricular assist device therapy: two comparable options in end-stage heart failure? Clin Cardiol 2013;36:378-82. [PubMed]
- Sandau KE, Hoglund BA, Weaver CE, et al. A conceptual definition of quality of life with a left ventricular assist device: results from a qualitative study. Heart Lung 2014;43:32-40. [PubMed]
- Maciver J, Ross HJ. Quality of life and left ventricular assist device support. Circulation 2012;126:866-74. [PubMed]





