Technique for minimizing and treating driveline infections
Introduction
Heart failure continues to be a leading cause of death, affecting over seven million people (1). In the United States, 500,000 new cases of heart failure are diagnosed each year (2). Among these patients, there has been a trend towards younger age at presentation. Nearly 30% of those admitted with congestive heart failure in 2010 were under the age of 65 (an increase from 23% in 2000) (3). While medical therapy improves survival overall, mortality after a single heart failure admission still approaches 33% (1). Heart transplantation has traditionally been the standard of care for advanced heart failure. However, the availability of donor organs has not been sufficient to meet the demands of this population.
Left ventricular assist devices (LVADs) have been shown to improve survival compared to medical management in patients with advanced heart failure (4-6). As such, LVADs are currently indicated both as bridge to transplantation as well as for destination therapy. With these indications, the number of LVADs implanted each year continues to rise. In 2010, over 1,500 implanted LVADs were implanted in the USA alone (7). Recent technological advancements have improved device durability. With fewer device associated morbidities (8), LVADs are now being implanted in relatively healthier populations. These patients are subsequently living longer, increasing the overall prevalence of patients with LVAD therapy.
As the population of patients living with an LVAD grows, there is increased awareness of long term complications that can arise with the device. Some of the most common complications are associated with the transcutaneous driveline. The transcutaneous driveline is necessary to power the LVAD and, although this technology has improved over the years in terms of smaller size and increased durability, driveline complications continue to develop in up to 20% of all LVADs implanted (9).
The LVAD driveline can be cumbersome for the patient and is prone to trauma and infection (10). Driveline infections can occur at any time after device insertion, with the majority presenting more than 30 days after implantation. Late driveline infections have been associated with driveline trauma and longer duration of support (11). The morbidity associated with a driveline infection is substantial and can include hospitalization, need for reoperation, increased risk of stroke, and delay in transplantation (12). In some studies, there has been a demonstrated trend toward decreased survival among those patients who present with a driveline infection (13).
The implications of a driveline infection necessitate a focused approach to driveline management from the time of surgery until well after discharge. For patients who present with a driveline infection, early recognition and treatment is important. Many institutions have evolved new surgical techniques for LVAD implantation as well as specialized postoperative management of the driveline. In this paper, we describe the evolution of our multidisciplinary approach for minimizing and treating driveline infections at the University of Virginia (Video 1).
Technique
Evolution of driveline management
Driveline management at our institution has evolved since the first HeartMate II (Thoratec) LVAD was implanted in January 2009. From the outset, all aspects of driveline management, from surgical implantation to postoperative care, were continually evaluated using a multidisciplinary approach. Careful documentation, including photographs of all driveline exit sites, allowed for objective review of each case. Specific intraoperative changes were made over the years that included burying the driveline velour below the skin edge and modifying the anchoring technique (Figure 1). Concomitantly, significant efforts were made to standardize postoperative patient education related to the driveline care and early management of driveline infections. Our current protocol is described below.
Surgical technique
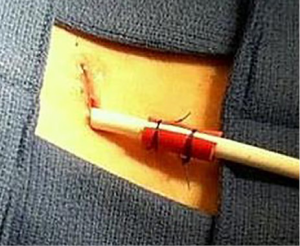
All patients receive prophylactic perioperative antibiotics (either cefazolin or vancomycin) which are continued for 48 hours after surgery. After the LVAD inflow and outflow anastomoses are completed, a 1.5 cm transverse incision is made 3 cm below the costal margin in the right upper quadrant at the mid-axillary line. Minimal bovie cautery is used to ensure hemostasis at the exit site. The blunt tunneling tool is then used to pass the driveline through the abdominal wall in a single puncture technique. The driveline velour is left completely buried 1 cm below the skin edge. The tissue around the driveline is then closed using a single layer 4-0 monocryl suture and sealed with a cyanoacrylate tissue adhesive. The driveline is further secured using two 2-0 prolene sutures around a piece of 28 French red rubber catheter that is cut longitudinally, and placed around the proximal driveline 2 cm from the exit site (Figure 2). Loosely tying the prolene above the skin prevents necrosis, and facilitates dressing of the driveline and subsequent suture removal. Careful attention is taken to ensure that the driveline site is completely isolated from the chest tubes and other dressings.
Inpatient education
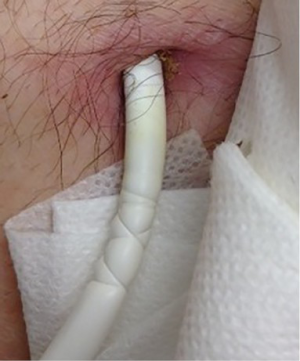
Strict attention to driveline cleanliness is ensured from post-operative day zero. Nursing teams have been instructed on twice daily cleaning routines which include the use of a mask and sterile gloves while cleaning the site. Once drainage from the exit site slows, the patient transitions to once daily driveline care. A driveline management pack has been created which contains all of the necessary equipment for a sterile dressing change. The patient and at least one family member are instructed on the driveline exit site management (DLES) technique (Figure 3).

Inpatient education about driveline care includes topical management as well as careful attention to manipulation of the controller. Dropping the controller and subsequent traction injury is one of the more common injuries to the driveline exit site in the immediate postoperative period. Patients also receive monitored training on showering technique to ensure that the driveline remains protected. On average, our patients receive 2-3 weeks of in-house training with family members before their discharge home.
Outpatient management
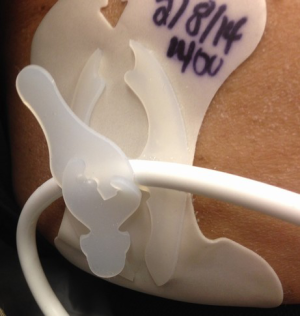
Patients are discharged with the same driveline management pack used in the hospital as well as written instructions on driveline care. Careful instructions are also provided to the patient in the event of trauma to the driveline. This includes an additional cleansing protocol of topical povidone-iodine twice a day and notification of their outpatient coordinator. The anchoring sutures that were placed at the time of surgery are cut and the red rubber catheter removed at one month postoperatively, at which time a new anchoring device is provided to the patient (Figure 4).
Driveline exit sites are recorded with photographs at each outpatient follow up visit. If the patient has any concern regarding exit site or driveline management, they are instructed to call the clinic and are often able to send a representative photo to the outpatient coordinator for review.
Diagnosing a driveline infection
If there is clinical concern for a driveline infection, including drainage or surrounding erythema, the patient is brought to clinic for evaluation. Culture swabs are obtained from the surrounding area along with complete blood counts (CBC) and blood culture studies. The driveline is carefully inspected for areas of trauma and the LVAD examined for any abnormal activity that might indicate driveline injury. If there is concern for deep infection, the workup also includes a CT scan.
Driveline infection management
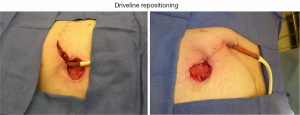
If culture results are positive or there is clinical concern for an infection, antibiotic therapy is initiated early. The most commonly isolated organisms are gram positive cocci and pseudomonas. For localized infections at the exit site, the patient is closely monitored during this time. Failure to clear the infection or evidence of a deeper abscess pocket warrants driveline repositioning or consideration of LVAD exchange. We have had success at repositioning the driveline by excising the tissue circumferentially around the exit site and unroofing the driveline to the point where it is fully incorporated (Figure 5).
If the infection extends to the pump pocket, an LVAD exchange is performed. The patient is placed on peripheral bypass and access to the LVAD pump is obtained through an extended subcostal incision. The pump is exchanged and the new driveline tunneled to a clean site on the abdomen. Antibiotic beads are left in the pump pocket to provide added defense against residual bacteria. Patients who are transplant candidates are transplanted as soon as an organ becomes available.
Destination therapy patients with driveline infections represent a concerning population, as deep infections can be difficult to clear even with pump exchange. In some instances, exchange of the entire LVAD including the inflow and outflow graft has been successful. Additionally, for those patients who refused pump exchange or were otherwise unable to undergo the operation, wide debridement of the pump pocket and driveline with the institution of antibiotic beads and negative pressure dressing therapy has been successful in suppressing the infection. These patients are also placed on chronic suppressive antibiotic therapy.
Results
Since 2009, 157 continuous flow LVADs have been implanted in 136 patients at the University of Virginia. Of these, 119 have been discharged and followed as an outpatient. Eighteen patients (15%) have had a documented driveline infection. In 2011, there was a significant upturn in the number of outpatients who had a driveline infection (21%). A multidisciplinary review was performed and new strategies have been employed to bring this number to zero. In the fall of 2011, the velour on the driveline was buried beneath the skin during implantation. Protocols were also created and implemented regarding driveline cleansing in the postoperative period. In 2012, there were no documented driveline infections. This year, the prepackaged kits containing the necessary sterile supplies were introduced. At the end of 2013, the red rubber driveline anchor was implemented at the time of surgery. Ultimately, between January 2012 and March 2014, we had zero driveline infections in our outpatient LVAD population (Figure 6).

Discussion
As patients live longer with LVAD therapies, they are more likely to experience complications associated with the transcutaneous driveline. In this paper, we describe the evolution of driveline management at a single institution which has significantly reduced the incidence of driveline infections. Other LVAD centers have reported on their strategies for driveline management, which include extensive broad spectrum antibiotics (14), extended tunneling of the driveline (15), and modified anchoring techniques. The conclusions from these experiences are that careful intraoperative technique and long term driveline immobilization are important in reducing the incidence of driveline trauma and infection.
Patient education is increasingly important as patients live longer with the devices and return to their outpatient communities. In a recent study by Sharma and colleges, the median time to development of a driveline infection was 182 days (2). The group most at risk for this complication is those patients who received an LVAD as destination therapy. These findings have been echoed in other studies and may be a reflection of a sicker and more elderly population as well as the increased duration of therapy (13).
In the occasion that a patient does develop a driveline infection, early identification is crucial. Up to 30% of driveline infections can be related to some form of trauma (e.g., dropping the controller) which can create a minor but significant disruption in the patient/prosthetic barrier. Increased topical cleansing and early notification to the LVAD coordinator can help to mitigate any potential infection. The majority of patients who do present with a concern for driveline infection have a superficial cellulitis surrounding the driveline exit site. Prompt initiation of culture directed antibiotics can be successful in clearing the infection. The reported experience of LVAD centers varies widely with regard to the success of antibiotics alone. Some centers report that 70% of patients with a driveline infection were able to be treated with antibiotics alone (2), whereas other groups report the need for surgical intervention in 60% of the cases (13).
Superficial exit site infections can extend to involve the distal driveline or even pump pocket. In these cases, aggressive surgical debridement is indicated. Several reports have demonstrated success with repositioning of the driveline when the infection is isolated to the exit site (16). Negative pressure dressings and local depot antibiotics have been used for pocket infections in addition to full pump exchange (17,18). Patients who have chronic infections of the LVAD have been successfully transplanted with subsequent eradication of the infection. However, there is some data to suggest that the overall rates of transplantation are reduced in this population (13). For patients with destination therapy LVADs, the treatment options can be limited. Suppressive antibiotic therapy is used if there has been evidence of a device related infection and the device is unable to be explanted.
Conclusions
In summary, the number of continuous flow LVADs implanted for management of heart failure continues to grow. As long as LVADs are dependent on an external power source, attention to detail regarding the driveline is crucial. While driveline infections will inevitably occur, we have been able to significantly reduce our driveline site infection rate through a multidisciplinary critical review process and use of standardized protocols. The implementation of the red rubber stabilization technique at the time of surgery has improved the ease of driveline care while maintaining stability of the driveline in the immediate postoperative period. The creation of a driveline management pack and improved patient education, including povidone-iodine dressing care with any trauma, has significantly reduced driveline infections since 2012. Early recognition and aggressive surgical approaches to driveline infection is essential to minimize associated morbidities.
Acknowledgements
The authors had full control of the methods used, outcome parameters and results, analysis of data and production of the written work.
Disclosures: There were no external sources of funds used to support this work. This manuscript has not been submitted for publication elsewhere.
References
- Miller LW. Left ventricular assist devices are underutilized. Circulation 2011;123:1552-8; discussion 1558. [PubMed]
- Sharma V, Deo SV, Stulak JM, et al. Driveline infections in left ventricular assist devices: implications for destination therapy. Ann Thorac Surg 2012;94:1381-6. [PubMed]
- CDC/NHCS. National Hospital Discharge Survey 2000-2010.
- Rose EA, Gelijns AC, Moskowitz AJ, et al. Long-term use of a left ventricular assist device for end-stage heart failure. N Engl J Med 2001;345:1435-43. [PubMed]
- Miller LW, Pagani FD, Russell SD, et al. Use of a continuous-flow device in patients awaiting heart transplantation. N Engl J Med 2007;357:885-96. [PubMed]
- Rogers JG, Butler J, Lansman SL, et al. Chronic mechanical circulatory support for inotrope-dependent heart failure patients who are not transplant candidates: results of the INTrEPID Trial. J Am Coll Cardiol 2007;50:741-7. [PubMed]
- Birks EJ. A changing trend toward destination therapy: are we treating the same patients differently? Tex Heart Inst J 2011;38:552-4. [PubMed]
- Slaughter MS, Rogers JG, Milano CA, et al. Advanced heart failure treated with continuous-flow left ventricular assist device. N Engl J Med 2009;361:2241-51. [PubMed]
- Goldstein DJ, Naftel D, Holman W, et al. Continuous-flow devices and percutaneous site infections: clinical outcomes. J Heart Lung Transplant 2012;31:1151-7. [PubMed]
- Holman WL, Rayburn BK, McGiffin DC, et al. Infection in ventricular assist devices: prevention and treatment. Ann Thorac Surg 2003;75:S48-57. [PubMed]
- Zierer A, Melby SJ, Voeller RK, et al. Late-onset driveline infections: the Achilles’ heel of prolonged left ventricular assist device support. Ann Thorac Surg 2007;84:515-20. [PubMed]
- Gordon SM, Schmitt SK, Jacobs M, et al. Nosocomial bloodstream infections in patients with implantable left ventricular assist devices. Ann Thorac Surg 2001;72:725-30. [PubMed]
- Topkara VK, Kondareddy S, Malik F, et al. Infectious complications in patients with left ventricular assist device: etiology and outcomes in the continuous-flow era. Ann Thorac Surg 2010;90:1270-7. [PubMed]
- Hieda M, Sata M, Seguchi O, et al. Importance of early appropriate intervention including antibiotics and wound care for device-related infection in patients with left ventricular assist device. Transplant Proc 2014;46:907-10. [PubMed]
- Schibilsky D, Benk C, Haller C, et al. Double tunnel technique for the LVAD driveline: improved management regarding driveline infections. J Artif Organs 2012;15:44-8. [PubMed]
- Walter V, Stock UA, Soriano-Romero M, et al. Eradication of a chronic wound and driveline infection after redo-LVAD implantation. J Cardiothorac Surg 2014;9:63. [PubMed]
- Baradarian S, Stahovich M, Krause S, et al. Case series: clinical management of persistent mechanical assist device driveline drainage using vacuum-assisted closure therapy. ASAIO J 2006;52:354-6. [PubMed]
- Garatti A, Giuseppe B, Russo CF, et al. Drive-line exit-site infection in a patient with axial-flow pump support: successful management using vacuum-assisted therapy. J Heart Lung Transplant 2007;26:956-9. [PubMed]