Sutureless Aortic Valve Replacement International Registry (SU-AVR-IR): design and rationale from the International Valvular Surgery Study Group (IVSSG)
Abstract
Background: Sutureless aortic valve replacement (SU-AVR) is an innovative approach which shortens cardiopulmonary bypass and cross-clamp durations and may facilitate minimally invasive approach. Evidence outlining its safety, efficacy, hemodynamic profile and potential complications is replete with small-volume observational studies and few comparative publications.
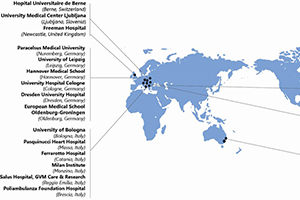
Methods: Minimally invasive aortic valve surgery and high-volume SU-AVR replacement centers were contacted for recruitment into a global collaborative coalition dedicated to sutureless valve research. A Research Steering Committee was formulated to direct research and support the mission of providing registry evidence warranted for SU-AVR.
Results: The International Valvular Surgery Study Group (IVSSG) was formed under the auspices of the Research Steering Committee, comprised of 36 expert valvular surgeons from 27 major centers across the globe. IVSSG Sutureless Projects currently proceeding include the Retrospective and Prospective Phases of the SU-AVR International Registry (SU-AVR-IR).
Conclusions: The global pooling of data by the IVSSG Sutureless Projects will provide required robust clinical evidence on the safety, efficacy and hemodynamic outcomes of SU-AVR.
Cover