A meta-analysis of deep hypothermic circulatory arrest versus moderate hypothermic circulatory arrest with selective antegrade cerebral perfusion
Introduction
The optimal selection of cerebral protection strategies is of critical in aortic arch surgery. A fundamental component of this has been deep hypothermic circulatory arrest (DHCA), which has in recent years been supplemented by retrograde cerebral perfusion (RCP) and then selective antegrade cerebral perfusion (SACP). The implementation of DHCA permits a bloodless field of operation as well as reducing systemic metabolic demands, while cerebral perfusion further fulfils residual cerebral metabolism (Figure 1) (1).
The role of temperature control and adjunctive perfusion in hypothermic circulatory arrest is of vital important. Earlier protocols have utilized temperatures as low as 14 °C (2), with the belief that lower temperatures alone will diminish cerebral metabolic demands sufficiently, so as to permit arch surgery. In recent years, surgeons have begun to question the limitations of using deep hypothermia. Implementation of adjunctive SACP has been sufficient to mollify the slight increase in brain metabolism caused by warmer hypothermic temperatures (3). While deep hypothermia alone is indeed sufficient to reduce brain metabolism and will increase safe circulatory arrest time (4), it has been associated with adverse systemic outcomes such as coagulopathy, renal and respiratory failure and increased inflammatory response (5,6). As such, moderate hypothermic circulatory arrest (MHCA) with SACP has been favoured in several institutions (7-9). The present meta-analysis aims to assess the clinical benefits of traditional DHCA strategy versus modern MHCA and SACP neuroprotective techniques.
Methods
Literature search strategy
Electronic searches were performed using Ovid Medline, PubMed, Cochrane Central Register of Controlled Trials (CCTR), Cochrane Database of Systematic Reviews (CDSR), ACP Journal Club, and Database of Abstracts of Review of Effectiveness (DARE) from their date of inception to January 2013. To achieve maximum sensitivity of the search strategy and to identify all studies, we combined the terms “cerebral perfusion” or “antegrade” or “circulatory arrest” and “aortic arch” as either key words or MeSH terms. The reference lists of all retrieved articles were reviewed for further identification of potentially relevant studies. All identified articles were systematically assessed using the inclusion and exclusion criteria. Expert academic cardiothoracic surgeons from 10 international aortic institutions formed the expert advisory panel and were asked whether they knew any unpublished data.
Selection criteria
Eligible comparative studies for the present meta-analysis included those in which patient cohorts underwent DHCA, or MHCA with SACP, for aortic arch surgery. Deep hypothermia is defined as 14.1-20 °C, while moderate hypothermia ranges from 20.1-28 °C, as per Expert Consensus by Yan et al. (Table 1) (10). Studies that did not include primary endpoint of stroke, or those whose primary patient set had a mean or median age less than 18 years old, were excluded. All publications were limited to those involving human subjects and in the English language. Abstracts, case reports, conference presentations, editorials, and expert opinions were excluded. Review articles were omitted because of potential publication bias and duplication of results. Studies that included fewer than 10 patients were also excluded.

Full table
Primary endpoints assessed included permanent neurological deficit (PND), temporary neurological deficit (TND), and perioperative death. PND is defined as either stroke and/or coma and somnolence, while TND is defined as postoperative confusion, obtundation, agitation, delirium, focal deficits resolving within 24-72 hours, seizures or psychosis, or transient ischemic attacks. Perioperative death is defined as death occurring within 30 days or within the same hospital stay. Secondary endpoints included postoperative renal failure and reoperation for bleeding.
Data extraction and critical appraisal
All data were extracted from article texts, tables and figures. When insufficient data were available from publications, corresponding authors were contacted to provide additional records. Two investigators (D.H.T. and B.W.) independently reviewed each retrieved article. Discrepancies between the two reviewers were resolved by discussion and consensus. The final results were confirmed by the senior investigator (T.D.Y.).
Statistical analysis
Meta-analysis was performed by combining the results of reported PND, TND, mortality, renal failure and reoperation for bleeding. The odds ratio (OR) was used as a summary statistic. In the present study, both fixed- and random-effect models were tested. In a fixed-effects model, it was assumed that treatment effect in each study was the same, whereas in a random-effects model, it was assumed that there were variations between studies. The calculated ratios thus had more conservative value (11). χ2 tests were used to study heterogeneity between trials. I2 statistic was used to estimate the percentage of total variation across studies owing to heterogeneity rather than chance. An I2 value of greater than 50% was considered substantial heterogeneity. If there was substantial heterogeneity, the possible clinical and methodologic reasons for this were explored qualitatively. In the present meta-analysis, the results using the random-effects model were presented to take into account the possible clinical diversity and methodologic variation between studies. Specific analyses considering confounding factors were not possible because raw data were not available.
Evidence of publication bias was sought using the methods of Egger et al. (12) and Begg et al. (13). Contour-enhanced funnel plot was performed to aid in interpreting the funnel plot (14). If studies appear to be missing in areas of low statistical significance, then it is possible that the asymmetry is due to publication bias. If studies appear to be missing in areas of high statistical significance, then publication bias is a less likely cause of funnel asymmetry. Intercept significance was determined by the t-test suggested by Egger et al. P<0.05 was considered representative of statistically significant publication bias.
All P values were 2-sided. All statistical analysis was conducted with Review Manager Version 5.2.1 (Cochrane Collaboration, Software Update, Oxford, United Kingdom), or STATA version 11.0 (Stata Corporation, College Station, TX).
Results
Quantity of studies
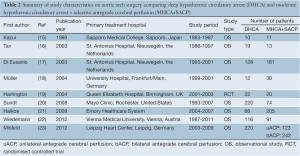
A total of 498 references were identified through 6 electronic database searches. After exclusion of duplicate or irrelevant references, 233 potentially relevant articles were retrieved. After detailed evaluation of these articles, 5 studies remained for assessment. Manual search of reference lists yielded 3 new studies. Of the 8 studies included for the present meta-analysis, one study was a randomized controlled trial (15), while the remaining were observational comparative studies (16-22). A large single-center observational study that specifically examined deep hypothermia at 22±2 °C versus moderate hypothermia with SACP was also included to better represent the available data from the literature (23). The significance of results is not affected by the inclusion of this study.
The study characteristics are summarized in Table 2. In these 9 studies, 1783 patients underwent aortic arch surgery, of whom DHCA were employed in 813 patients and MHCA+SACP used in 970 patients.
Full table
Baseline characteristics
Both comparative groups displayed similar baseline characteristics. Median age ranged from 55-65 years in DHCA group, and 58-66 years in MHCA+SACP group. Even distributions of male and female patients were observed; male patients undergoing DHCA alone ranged from 40-76%, compared to 51-73% of MHCA+SACP cohort, with no overall difference (64% vs. 66% males overall; OR, 1.03; 95% CI, 0.79-1.39; P=0.86). No other risk factors were consistently reported.
Frequency of acute dissection cases was similar in both DHCA and MHCA+SACP groups (48% vs. 44%; OR, 1.71; 95% CI, 0.91-3.19; P=0.09), with two studies only examining patients with acute type A dissection (17,22). Heterogeneity reporting of the extent of arch surgery precluded its analysis, although there existed a general trend for more complex operations in MHCA+SACP groups.
Circulatory arrest temperatures ranged from 15-20 °C for DHCA patients and 22-25 °C for MHCA+SACP patients. Significant variations in sites of temperature measurement were reported, including the nasopharynx (16,17,19,20), esophagus (22), rectum (16,19), bladder (20), or core (20,21). Either unilateral (19-22), or bilateral (15-18) SACP was employed. A summary of surgical protocols is presented in Table 3.

Full table
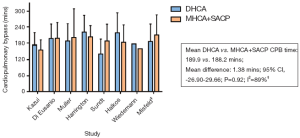
Cardiopulmonary bypass time was comparable between DHCA and MHCA+SACP (189.9 vs. 188.2 min; mean difference, 1.4 min; 95% CI, -26.9-29.7; P=0.92) (Figure 2). Time required for cooling was significantly longer in DHCA group (57 vs. 37 min; mean difference, 19.3 min; 95% CI, 4.6-34.0; P=0.01), although this was only reported statistically in two studies (15,16). Other variables reported included: circulatory arrest time (15,16,19-22), SACP time (15,16), cardiac ischemic time (18,19), cardiac arrest time (16) and total cerebral protection time (15,18). However, the lack of consistency in reporting precluded statistical analysis.
Neurological outcomes
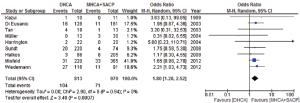
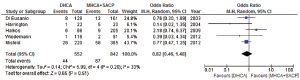
PND, reported in all 9 studies (15-23), was significantly higher in the DHCA group as compared to MHCA+SACP group (12.8% vs. 7.3%; OR, 1.80; 95% CI, 1.28-2.52; P=0.0007; I2=0%) (Figure 3).
In contrast, no significant difference was observed for TND, between DHCA and MHCA+SACP when reported (8.0% vs. 10.3%; OR, 0.82; 95% CI, 0.46-1.48; P=0.51; I2=33%) (15,18,21-23) (Figure 4).
Mortality outcomes
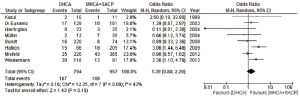
Eight studies presented details on perioperative mortality (15,16,18-23), with no significant difference observed between DHCA and MHCA+SACP groups (13.5% vs. 11.1%; OR, 1.39; 95% CI, 0.88-2.20; P=0.15, I2=43%) (Figure 5).
Morbidity outcomes
In studies that reported post-operative renal failure (16,20-23), both DHCA and MHCA+SACP groups had comparable outcomes (13.3% vs. 12.6%; OR, 1.36; 95% CI, 0.74-2.49; P=0.32; I2=40%). Reoperation for bleeding was similarly comparable between both groups when reported (10.9% vs. 13.3%; OR, 0.85; 95% CI, 0.43-1.69; P=0.65; I2=62%) (16,18,20,23).
Publication bias
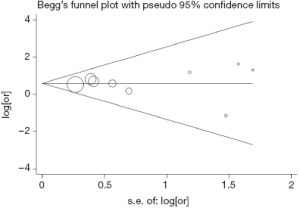
Begg’s funnel plot and Egger’s test were performed to assess publication bias in the literature. All 9 included studies yielded a Begg’s test score of P=0.917 and an Egger’s test score of P=0.902, meanwhile inspection of the contour-enhanced funnel plot (Figure 6) shows absence of publication bias in all 9 studies. These results suggest that publication bias was not an influencing factor.
Discussion
Since Griepp and associates first described the implementation of hypothermic circulatory arrest in aortic arch surgery in 1975 (2), pioneering cardiac surgeons have continued to push the boundary of arch surgery; adjunct RCP has mostly been superseded by antegrade cerebral perfusion. Perfusion techniques vary from direct cannulation of supra-aortic vessels (24), and to axillary cannulation (25). More recently, the role of deep hypothermia alone has been questioned, and at the same time the necessity of maintaining extremely low cerebral metabolic rates has being partly negated by the implementation of SACP. Deep hypothermic temperatures have been reported to be associated with hypothermia-induced coagulopathy, cerebral microvasculature endothelial dysfunction, neuronal damage, increased systemic inflammatory response, and increased risk of organ dysfunction (5,6,26-28). In response to this, several institutions have implemented warmer temperatures for circulatory arrest. The present meta-analysis aimed to assess the benefits of MHCA and SACP over traditional methods of using DHCA alone.
The raison d’etre for using SACP has been its tolerance for longer periods of safe circulatory arrest. SACP maintains near-physiological cerebral circulation with homogenous distribution of blood throughout the capillary beds, albeit with a non-pulsatile and hypothermic flow pattern (29). Postoperative quality of life is also generally increased compared to DHCA alone irrespective of duration of circulatory (30). While DHCA alone is sufficient for 30-40 minutes of circulatory arrest (4,31,32), adjunctive SACP may extend this period to 80 minutes (32). However, operations that necessitate these prolonged arrest periods are by nature more complex, and thus carry greater risk. Despite this potential bias of baseline characteristics against the MHCA+SACP cohort, comparable results, such as mortality, were observed as compared to traditional DHCA, suggesting the non-inferiority of MHCA+SACP despite the added complexity of arch surgery. These results are in line with the findings of a Best Evidence Topic study examining the benefits of SACP, which concluded that SACP is superior as an adjunct compared to hypothermic circulatory arrest alone, although the authors underscored the weakness of the available clinical evidence (33).
The present meta-analysis indicates that MHCA+SACP significantly reduces the risk of post-operative stroke. It has been argued that the majority of strokes after proximal aortic arch surgery are embolic in nature (21). However, our results show that studies (15-18,23) that perfuse even through direct cannulation of supraaortic vessels (with an alleged greater chance of dislodging atherothrombi), still maintain significantly better permanent neurologic outcomes than using DHCA alone. This might be attributed to antergrade cerebral perfusion, which provides continuous blood supply to the brain, and methodical care during cannulation. Even in complex aortic arch surgery requiring longer neuroprotection, whereby total brain exclusion time is strongly associated with stroke (32,34), appropriate use of SACP is considered an acceptable strategy. Several authors advocate careful deairing techniques to minimise introduction of foreign debris into cerebral circulation, dissection of arch vessels distant from atherosclerotic plaques, or avoiding cannulation of arch vessels (21,35,36).
Whereas PNDs are localised events, TNDs are subtle and diffuse global injuries undetectable by conventional imaging methods, and are associated with inadequate perfusion. Indeed, Merkkola and associates reported that unilateral cerebral perfusion might not sufficiently supply the contralateral hemisphere in 17% of patients (37). However, while the circle of Willis is only reported to be complete in 42-47% of patients (38,39), Urbanski and colleagues argued that anatomic completeness of the circle of Willis did not correlate with insufficient brain protection during unilateral cerebral perfusion (40). Nevertheless, several institutions advocated bilateral cerebral perfusion when preoperative assessments indicated poor vascular patency (41,42). In the present meta-analysis, no significant difference was observed between either DHCA or MHCA+SACP, although four studies that utilised bilateral SACP presented favourable TND results (15,18,22,23), in contrast to results from the single study that utilised unilateral perfusion (21). However, it needs to be noted that significant variations exist in the definition and assessment of TND, which has the potential to confound these findings.
The impetus for using MHCA and SACP over DHCA alone has been the potential complications associated with deep hypothermic temperatures. Single institutional studies have demonstrated favourable systemic outcomes using MHCA. Halkos and colleagues reported significant reduction in the incidence of renal failure in patients undergoing MHCA and SACP compared to DHCA (21), while Di Eusanio and associates further observed that renal dysfunction was significantly more common in the DHCA cohort (18).
Despite these studies, the general impression that deep hypothermia is associated with greater systemic morbidities has not been conclusively validated clinically. Rather, inferences have often been extrapolated from laboratory experiments or ancillary studies, as well as from the axiomatic belief that disturbance from normal physiological parameters is undesirable. Our overall analysis demonstrated no significant impact of using MHCA and SACP on reducing renal failure, and comparable incidence for reoperation for bleeding. However, it was noted that systemic outcomes are infrequently and inconsistently reported, thus limiting the evaluation of the rationalization behind MHCA. Furthermore, the evolution of arch surgical techniques may have overcome the perceived disadvantages of DHCA; for example, CPB time in the DHCA group in the present is not significantly different from the MHCA+SACP cohort, obviating concerns of extended surgery due to time required for rewarming.
While the literature is replete with retrospective reports of arch surgery, no conclusive randomized controlled trial or registry-based study has so far evaluated comparisons of operative techniques. The single randomized study included in this meta-analysis was not sufficiently powered to evaluate clinical outcomes (15). The impetus falls upon high-volume aortic centers to conduct large scale RCTs to better appraise the benefits of MHCA and SACP. Failing this, a comprehensive international multi-centric database needs to be established, of sufficient robustness, to adequately stratify patient characteristics, arch pathologies, and operative strategies. Analysis of cerebral protection reported in existing registries, such as the International Registry of Acute Aortic Dissection (43) and the German Registry for Acute Aortic Dissection Type A (GERAADA) (44), may be informative.
A limitation of the present meta-analysis is the heterogeneous nature of arch pathologies and variations in operative strategies. Two studies examined only acute type A dissection patients (17,22), which is an independent predictor of stroke (23). Variations in operative technique, study design, and reporting have precluded comparison of the true duration of cerebral or systemic circulatory arrest, as well as limiting stratification by the extent of arch repair, which may have influenced factors such as CPB time. Inconsistent reporting of systemic outcomes, such as renal/hepatic/pulmonary dysfunction, bleeding, and spinal injury, has further limited our assessment.
In conclusion, the present meta-analysis demonstrates that MHCA with SACP significantly reduces incidence of stroke compared to using DHCA alone. Comparable results in TND and mortality demonstrate the non-inferiority of MHCA+SACP, despite a trend for its use in more complex surgical cases. More consistent reporting of outcomes and studies with higher levels of evidence is encouraged to further evaluate the benefits of moderate hypothermia and cerebral perfusion.
Acknowledgements
The authors would like to thank Dr. Ping Zhan for statistical analysis of publication bias.
Disclosure: The authors declare no conflict of interest.
References
- Ziganshin B, Elefteriades J. Deep hypothermic circulatory arrest. Ann Cardiothorac Surg 2013. doi: 10.3978/j.issn.2225-319X.2013.01.05. [Epub ahead of print].
- Griepp RB, Stinson EB, Hollingsworth JF, et al. Prosthetic replacement of the aortic arch. J Thorac Cardiovasc Surg 1975;70:1051-63.
- Stecker MM, Cheung AT, Pochettino A, et al. Deep hypothermic circulatory arrest: I. Effects of cooling on electroencephalogram and evoked potentials. Ann Thorac Surg 2001;71:14-21.
- McCullough JN, Zhang N, Reich DL, et al. Cerebral metabolic suppression during hypothermic circulatory arrest in humans. Ann Thorac Surg 1999;67:1895-9.
- Livesay JJ, Cooley DA, Reul GJ, et al. Resection of aortic arch aneurysms: a comparison of hypothermic techniques in 60 patients. Ann Thorac Surg 1983;36:19-28.
- Kamiya H, Hagl C, Kropivnitskaya I, et al. The safety of moderate hypothermic lower body circulatory arrest with selective cerebral perfusion: a propensity score analysis. J Thorac Cardiovasc Surg 2007;133:501-9.
- Khaladj N, Shrestha M, Meck S, et al. Hypothermic circulatory arrest with selective antegrade cerebral perfusion in ascending aortic and aortic arch surgery: a risk factor analysis for adverse outcome in 501 patients. J Thorac Cardiovasc Surg 2008;135:908-14.
- Urbanski PP, Lenos A, Bougioukakis P, et al. Mild-to-moderate hypothermia in aortic arch surgery using circulatory arrest: a change of paradigm? Eur J Cardiothorac Surg 2012;41:185-91.
- Ueda T, Shimizu H, Ito T, et al. Cerebral complications associated with selective perfusion of the arch vessels. Ann Thorac Surg 2000;70:1472-7.
- Yan TD, Bannon PG, Bavaria J, et al. Consensus on hypothermia in aortic arch surgery. Ann Cardiothorac Surg 2013;2:163-168.
- DerSimonian R, Laird N. Meta-analysis in clinical trials. Control Clin Trials 1986;7:177-88.
- Egger M, Davey Smith G, Schneider M, et al. Bias in meta-analysis detected by a simple, graphical test. BMJ 1997;315:629-34.
- Begg CB, Mazumdar M. Operating characteristics of a rank correlation test for publication bias. Biometrics 1994;50:1088-101.
- Palmer T, Peters J, Sutton A, et al. Contour enhanced funnel plots for meta-analysis. Stata Journal 2008;8:242.
- Harrington DK, Walker AS, Kaukuntla H, et al. Selective antegrade cerebral perfusion attenuates brain metabolic deficit in aortic arch surgery: a prospective randomized trial. Circulation 2004;110:II231-6.
- Kazui T, Inoue N, Komatsu S. Surgical treatment of aneurysms of the transverse aortic arch. J Cardiovasc Surg (Torino) 1989;30:402-6.
- Tan ME, Dossche KM, Morshuis WJ, et al. Is extended arch replacement for acute type a aortic dissection an additional risk factor for mortality? Ann Thorac Surg 2003;76:1209-14.
- Di Eusanio M, Wesselink RM, Morshuis WJ, et al. Deep hypothermic circulatory arrest and antegrade selective cerebral perfusion during ascending aorta-hemiarch replacement: a retrospective comparative study. J Thorac Cardiovasc Surg 2003;125:849-54.
- Müller D, Fieguth HG, Wimmer-Greinecker G, et al. Neurologic outcome after surgery of the aortic arch: Comparison of deep hypothermic arrest, antegrade and retrograde cerebral perfusion. Indian J Thorac Cardiovasc Surg 2004;20:72-6.
- Sundt TM 3rd, Orszulak TA, Cook DJ, et al. Improving results of open arch replacement. Ann Thorac Surg 2008;86:787-96; discussion 787-96.
- Halkos ME, Kerendi F, Myung R, et al. Selective antegrade cerebral perfusion via right axillary artery cannulation reduces morbidity and mortality after proximal aortic surgery. J Thorac Cardiovasc Surg 2009;138:1081-9.
- Wiedemann D, Kocher A, Dorfmeister M, et al. Effect of cerebral protection strategy on outcome of patients with Stanford type A aortic dissection. J Thorac Cardiovasc Surg 2012. [Epub ahead of print].
- Misfeld M, Leontyev S, Borger MA, et al. What is the best strategy for brain protection in patients undergoing aortic arch surgery? A single center experience of 636 patients. Ann Thorac Surg 2012;93:1502-8.
- Kazui T, Washiyama N, Muhammad BA, et al. Improved results of atherosclerotic arch aneurysm operations with a refined technique. J Thorac Cardiovasc Surg 2001;121:491-9.
- Svensson LG, Blackstone EH, Rajeswaran J, et al. Does the arterial cannulation site for circulatory arrest influence stroke risk? Ann Thorac Surg 2004;78:1274-84; discussion 1274-84.
- Cooper WA, Duarte IG, Thourani VH, et al. Hypothermic circulatory arrest causes multisystem vascular endothelial dysfunction and apoptosis. Ann Thorac Surg 2000;69:696-702; discussion 703.
- Egerton N, Egerton WS, Kay JH. Neurologic Changes Following Profound Hypothermia. Ann Surg 1963;157:366-74.
- Wilde JT. Hematological consequences of profound hypothermic circulatory arrest and aortic dissection. J Card Surg 1997;12:201-6.
- Apostolakis E, Shuhaiber JH. Antegrade or retrograde cerebral perfusion as an adjunct during hypothermic circulatory arrest for aortic arch surgery. Expert Rev Cardiovasc Ther 2007;5:1147-61.
- Immer FF, Lippeck C, Barmettler H, et al. Improvement of quality of life after surgery on the thoracic aorta: effect of antegrade cerebral perfusion and short duration of deep hypothermic circulatory arrest. Circulation 2004;110:II250-5.
- Gega A, Rizzo JA, Johnson MH, et al. Straight deep hypothermic arrest: experience in 394 patients supports its effectiveness as a sole means of brain preservation. Ann Thorac Surg 2007;84:759-66; discussion 766-7.
- Hagl C, Ergin MA, Galla JD, et al. Neurologic outcome after ascending aorta-aortic arch operations: effect of brain protection technique in high-risk patients. J Thorac Cardiovasc Surg 2001;121:1107-21.
- Barnard J, Dunning J, Grossebner M, et al. In aortic arch surgery is there any benefit in using antegrade cerebral perfusion or retrograde cerebral perfusion as an adjunct to hypothermic circulatory arrest? Interact Cardiovasc Thorac Surg 2004;3:621-30.
- Matalanis G, Hata M, Buxton BF. A retrospective comparative study of deep hypothermic circulatory arrest, retrograde, and antegrade cerebral perfusion in aortic arch surgery. Ann Thorac Cardiovasc Surg 2003;9:174-9.
- Kazui T, Bashar AH, Washiyama N. Total aortic arch replacement and limited circulatory arrest of the brain. J Thorac Cardiovasc Surg 2005;129:1207-8.
- Di Eusanio M, Schepens MA, Morshuis WJ, et al. Brain protection using antegrade selective cerebral perfusion: a multicenter study. Ann Thorac Surg 2003;76:1181-8; discussion 1188-9.
- Merkkola P, Tulla H, Ronkainen A, et al. Incomplete circle of Willis and right axillary artery perfusion. Ann Thorac Surg 2006;82:74-9.
- Macchi C, Catini C, Federico C, et al. Magnetic resonance angiographic evaluation of circulus arteriosus cerebri (circle of Willis): a morphologic study in 100 human healthy subjects. Ital J Anat Embryol 1996;101:115-23.
- Krabbe-Hartkamp MJ, van der Grond J, de Leeuw FE, et al. Circle of Willis: morphologic variation on three-dimensional time-of-flight MR angiograms. Radiology 1998;207:103-11.
- Urbanski PP, Lenos A, Blume JC, et al. Does anatomical completeness of the circle of Willis correlate with sufficient cross-perfusion during unilateral cerebral perfusion? Eur J Cardiothorac Surg 2008;33:402-8.
- Kazui T, Inoue N, Yamada O, et al. Selective cerebral perfusion during operation for aneurysms of the aortic arch: a reassessment. Ann Thorac Surg 1992;53:109-14.
- Dossche KM, Schepens MA, Morshuis WJ, et al. Antegrade selective cerebral perfusion in operations on the proximal thoracic aorta. Ann Thorac Surg 1999;67:1904-10; discussion 1919-21.
- Hagan PG, Nienaber CA, Isselbacher EM, et al. The International Registry of Acute Aortic Dissection (IRAD): new insights into an old disease. JAMA 2000;283:897-903.
- Krüger T, Weigang E, Hoffmann I, et al. Cerebral Protection During Surgery for Acute Aortic Dissection Type AClinical Perspective Results of the German Registry for Acute Aortic Dissection Type A (GERAADA). Circulation 2011;124:434-43.





