Perspective on the cost-effectiveness of transapical aortic valve implantation in high-risk patients: Outcomes of a decision-analytic model
Background: The incremental cost-effectiveness of transapical transcatheter aortic valve implantation (TAVI)
is ill-defined in high-risk patients where aortic valve replacement (AVR) is an option, and has not been
ascertained outside a randomized controlled trial.
Methods: We developed a Markov model to examine the progression of patients between health states,
defined as peri- and post-procedural, post-complication, and death. The mean and variance of risks,
transition probabilities, utilities and cost of transapical TAVI, high-risk AVR, and medical management
were derived from analysis of relevant registries. Outcome and cost were derived from 10,000 simulations.
Sensitivity analyses further evaluated the impact of mortality, stroke, and other commonly observed
outcomes.
Results: In the reference case, both transapical TAVI and high-risk AVR and TAVI were cost-effective when
compared to medical management ($44,384/QALY and $42,637/QALY, respectively). Transapical TAVI
failed to meet accepted criteria for incremental cost-effectiveness relative to AVR, which was the dominant
strategy. In sensitivity analyses, the mortality rates related to the two strategies, the utilities post-AVR and
post-transapical TAVI, and the cost of transapical TAVI, were the main drivers of model outcome.
Conclusions: Transapical TAVI did not satisfy current metrics of incremental cost-effectiveness relative
to high-risk AVR in the reference case. However, it may provide net health benefits at acceptable cost in
selected high-risk patients among whom AVR is the standard intervention.
Key words: Submitted May 22. Accepted for publication Jun 21, 2012.
Introduction
Transcatheter approaches for management of aortic stenosis (AS) have revolutionized the treatment of AS in several patient populations. In patients of high, but not prohibitive, surgical risk, TAVI appears to provide similar outcomes to surgical aortic valve replacement (AVR), as demonstrated by the Placement of Aortic Transcatheter Valve (PARTNER) Cohort A (1). Analysis from this trial has determined that transfemoral, but not transapical, transcatheter aortic valve implantation (TAVI) is incrementally cost-effective relative to AVR.
The avoidance of bias in prospective randomized controlled trials have led these protocols to be considered the reference standard with respect to defining the effectiveness of therapies. However, concerns about external validity may arise if aspects of the trial differ from standard practice (2). As variations in the apparent effect of TAVI may be modulated by the prevalence and effect of comorbid disease, stricter inclusion/exclusion criteria in this circumstance may lead to potential inflation of relative benefit (3). Therefore, despite the risk of selection bias, prospective, registry data may provide insights additional to randomized studies. We sought to study the costeffectiveness of transapical TAVI using registry data, in order to provide better perspective into real-world clinical practice employing this technology.
Methods
Model development
Outcomes and costs attributed to transapical TAVI with the Edwards SAPIEN valve (Edwards Lifesciences, Irvine, CA) and tissue AVR for the management of high-risk patients with severe AS were evaluated in a decision-analytic model. This model was informed from registries of high-risk patients undergoing transapical TAVI and AVR, defined as a logistic EuroSCORE>15% and/or STS score>10% (Table 1) (4-23).
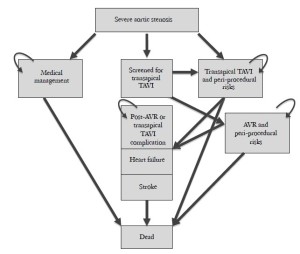
The passage of a hypothetical cohort of 10,000 patients through a number of health states that arose as consequence of transapical TAVI, AVR, or medical management (Figure 1) was assessed by Monte Carlo simulation using a Markov model (TreeAge Pro 2008, TreeAge, Williamstown, MA) (24). This approach allows estimation of quality-adjusted life years (QALY) and lifetime cost, and therefore calculation of the incremental cost-effectiveness ratio (ICER) of the technique with the most favorable outcome. Costs were estimated in 2012 US dollars; an adjustment to allow calculation of present value was performed using a discount rate of 5% per year in order to permit comparisons.
| Table 1 Characteristics of underlying groups (95% confidence intervals in parentheses, where applicable) | ||||
| AVR (4-11) | Transapical TAVI (12-13) | p | ||
|---|---|---|---|---|
| N | 1,544 | 2,786 | ||
| Age | 81.95 (81.59-82.31) | 81.20 (80.94-81.45) | 0.101 | |
| Female | 49.88% (43.13-56.64%) | 53.90% (46.62-61.17%) | 0.472 | |
| NYHA III-IV | 87.39% (71.13-100%) | 76.63% (70.44-82.83%) | 0.092 | |
| EF | 46.49% (44.47-48.52%) | 52.88% (51.35-54.42%) | 0.010 | |
| History of MI | 25.04% (4.69-45.39%) | 20.54% (8.36-32.71%) | 0.620 | |
| History of stroke | 19.17% (14.84-23.50%) | 13.25% (8.57-17.93%) | 0.110 | |
| History of PAD | 27.42% (12.86-41.98%) | 48.57% (39.52-57.62%) | 0.016 | |
| Diabetes | 22.30% (12.13-32.47%) | 27.18% (24.31-30.04%) | 0.335 | |
| Hypertension | 78.89% (64.84-92.93%) | 77.61% (74.21-81.01%) | 0.652 | |
| Renal failure | 23.84% (11.44-36.23%) | 16.78% (9.08-24.49%) | 0.514 | |
| Logistic EuroSCORE | 30.77% (26.36-35.18%) | 29.39% (26.59-31.83%) | 0.696 | |
| STS score | 13.26 (7.18-19.33) | 12.51 (10.77-14.26) | 0.843 | |
Health states, transitions and assumptions
The cycle length of the model (i.e. the interval at which patients changed health states) was set at 1 year, and the rest of life was simulated. Candidates for AVR (mean initial age of 80 years) entered the model with severe AS. The frequency of outcomes as well as transitions between health states, were obtained from the literature, using pooled estimates (Table 2). Transitions in operable patients who are medically managed were obtained from a registry that compared outcomes of patients offered intervention with AVR or transapical TAVI versus medical management (25). We assumed that only a proportion of patients that underwent work-up would proceed to transapical TAVI. The proportion of patients evaluated for transapical TAVI and then found unsuitable for implantation would include those converted to AVR after failed transapical TAVI implantation, based on the premise that the transapical implantation is being used as an alternative to standard AVR, rather than being in response to inoperability.
| Table 2 Transition probabilities and mortality rates (95% confidence intervals in parentheses, where applicable) | ||||
| AVR | Transapical TAVI | Medical management | ||
|---|---|---|---|---|
| Peri-operative events | ||||
| Death* | 9.76% (6.75-12.76%) (4-11) | 9.73% (7.51-11.95%) (12-23) | 15.40% (25) | |
| TAVI access site Complications | - | 2.53% (0.99-4.07%) (12-14,17-20,22,23) | - | |
| Stroke ‿Peri-operative | 3.78% (2.70-4.86%) (4-9,11) | 2.00 (1.37-2.62%) (12-15,17-23) | - | |
| Follow-up events | ||||
| Death | 24.21% (18.87-29.55%) (7-11) | 25.28 (21.06-29.51%) (12-18,20,21,23) | 55.10% (25) | |
| Heart failure | 11.30% (26) | 100% | ||
| Stroke ‿Post-operative | 2.10% (2.70-3.70%) (27) | 3.55% (3.42-3.68%) (12,14,17) | - | |
| Post-AVR/TAVI Complications** | 4.80% (4.10-5.60%) (27) | - | ||
| Reoperation | 0.24% (27) | 3.28% (3.19-3.37%) (12,14,17-19) | - | |
| Complication mortality*** | ||||
| TAVI access site complications | - | 25.80% (17) | - | |
| Post-AVR/TAVI complications | 25.89% (28) | - | ||
| Stroke - Peri-operative | style="border-bottom:#000000 solid 1px"11.00 (8.00-14.00%) (29) | - | ||
| *: Assumed to be 30-day mortality in medical management group; **, Complications include endocarditis, hemorrhage, valve thrombosis, and non-cerebral thromboembolism; ***, Annual mortality related to stroke and heart failure was assumed the same as that after AVR or TAVI | ||||
Overlapping health states were attributed the outcomes, utilities and costs of the worst state. For example, all patients have a substantial mortality, so this was not increased further in those with heart failure or stroke. Because of the high-risk status of patients in the medical management group, they were assumed to have heart failure. Likewise, the impact of stroke with regards to both cost and quality of life in the medical management group would be minor, so stroke was not assessed in the medical management group. Patients with stroke did not proceed to reoperation.
The risk of other post-AVR complications, including endocarditis, hemorrhage, valve thrombosis, and noncerebral thromboembolism, have not been adequately assessed in the transapical TAVI population, so we estimated them from linearized event rates in AVR recipients (27). Mortality related to post-AVR complications was derived from weighted averages from a meta-analysis of outcomes post-bioprosthetic AVR (28), and we assumed that as TAVI is a tissue valve, it would have a similar post-procedure complication rates.
Bleeding and vascular complications overlap, and were assessed as a single complication (12-14,17-20,22,23), with a mortality derived from the one-year outcomes of the SOURCE Registry (17). Access site complications were assumed to occur only in the first cycle post-transapical TAVI. Transapical TAVI patients undergoing valve replacement underwent only repeat TAVI (i.e. valve-invalve). The reoperation rate for tissue valves has previously been described to follow a Weibull distribution (30).
Health outcomes
This information is summarized in Table 3. Age-specific data were obtained from the Medical Expenditure Panel Survey (31), and utility was age-adjusted, declining by 0.3% per year of age. QALYs were calculated from utility weights, multiplied by the duration in each health state. Heart failure utility for functional classes II and III was derived from the Cardiovascular Outcomes Research Consortium (33), and subject to sensitivity analysis to compensate for the fact that our population was significantly older than those in the original report. Specific utilities for postoperative stroke are not well defined, so we used a utility for disabling stroke from a systematic review (34. Utilities regarding other post-AVR/TAVI complications are also not well established and were deemed equivalent to that of heart failure (33).
We applied a 10% reduction in utility for uncomplicated AVR from that predicted of an age-matched population (31). Post-transapical TAVI utility was attributed the status reported in the European PARTNER transcatheter heart valve study (14). We anticipated a decrement in utility from access site complications concordant with the added hospital length-of-stay in patients developing hemorrhage postcoronary artery bypass grafting (32). Utility of the medical management group was assessed as being equivalent to the QALY before transapical TAVI in the European PARTNER transcatheter heart valve study (14).
| Table 3 Utility values for each health state (±standard deviation) | ||||
| Heart failure | 0.67±0.01 (30) | |||
| Stroke | 0.46±0.01 (31) | |||
| Post-AVR | 0.69 based on 10% disutility (31) | |||
| Post-transapical TAVI | 0.66±0.43 (6) | |||
| Medical management | 0.59±0.30 (6) | |||
| Transapical TAVI access site complications | -1 QAL week (32) | |||
| Post-AVR/Transapical TAVI complications | 0.67±0.01* | |||
| *Assumed to be similar to that of NYHA functional class II and III | ||||
Cost information
Cost analysis was taken from the perspective of the healthcare funding body, assuming that the amount reimbursed to the provider is the cost of care. Cost information was mainly obtained from diagnostic related groups (DRGs), and Medicare payments for current procedural terminology (CPT) codes (Table 4). Costs associated with AVR were gathered from weighted national estimates from the Healthcare Cost and Utilization Project (HCUP) nationwide inpatient sample 2010 (36). We assumed that reimbursement for transapical TAVI would be analogous to AVR (39). Costs of additional imaging, such as computed tomographic angiography, were included in screening for transapical TAVI. The costs of heart failure, including the costs of procedures and provider visits (35), were also attributed to patients in the medical management group, based on their assumed utility status. Peri-operative stroke was assigned the costs for acute treatment and followup (36,37). The average cost of hemorrhage post-coronary artery bypass was used as a surrogate for that of access site complications related to transapical TAVI (32). A weighted average of the relative incidences of post-AVR and TAVI complications was used to assign average annual cost (38).
| Table 4 Costs associated with each health state | ||||
| Mean cost (US$/yr; ±standard deviation) | ||||
|---|---|---|---|---|
| Heart failure | Yearly costs after diagnosis: 10,832 (35) | |||
| Stroke | 14,155±453 (acute treatment) (36) |
|||
| TAVI | 49,106±1283 (33) |
|||
| AVR | 49,106±1283 (33) |
|||
| Medical management | Yearly costs after diagnosis: 10,832 (35) | |||
| TAVI access site complications | 12,302 (32) | |||
| Post-AVR/TAVI complications | Yearly costs: 300 (38) | |||
| *Follow-up costs for one outpatient visit; **Follow-up costs for transthoracic echocardiogram plus one outpatient visit | ||||
Analyses
Beta distributions (continuous probability distributions defined on an interval of 0 to 1) were assigned to probabilities and utility weights, and gamma distributions (continuous probability distributions potentially ranging from 0 to infinity) were assigned to costs based on standard errors derived from the associated literature. Means and 95% credible intervals (Bayesian confidence interval, 95% CI) were computed on the basis of 10,000 iterations. The willingness-to-pay (WTP) threshold of acceptable incremental cost-effectiveness was defined by an ICER of <$100,000/QALY. Probabilistic sensitivity analyses were performed from the Markov model, and one-way sensitivity analyses were performed to identify the critical sources of variation in the input data.
Results
Health outcomes and costs of high-risk AVR and transapical TAVI
Outcomes of transapical TAVI and AVR were superior to medical management, although lifetime costs of transapical TAVI and AVR exceeded those of medical management. The cost-effectiveness ratios of transapical TAVI and AVR were respectively $44,384/QALY and $42,637/QALY. In the reference case (age 80, transition probabilities and outcomes in Table 2, 50% suitability for transapical TAVI), transapical TAVI was dominated by AVR, because lifetime costs were slightly higher for transapical TAVI ($56,730 vs. $56,630), with slightly fewer QALYs from TAVI (1.66 vs. 1.70).
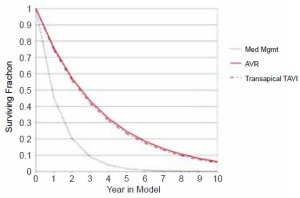
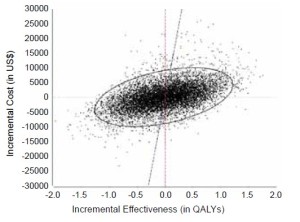
The anticipated survival curves from 10,000 simulations (Figure 2) showed both interventions to be superior to medical therapy. The incremental cost-effectiveness plot from simulation comparison of transapical TAVI relative to AVR (Figure 3) showed that at a WTP <$100,000/QALY gained, transapical TAVI is incrementally cost-effective relative to AVR in a minority (47%) of simulations.
Sensitivity analyses
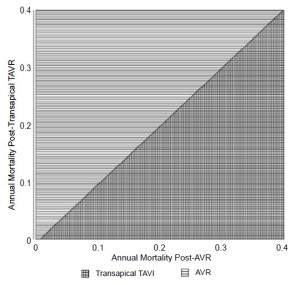
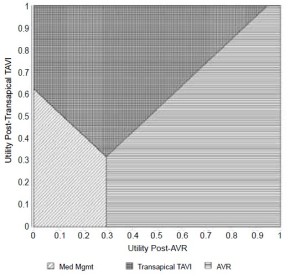
Analysis of each factor across a clinically plausible range was used in threshold analyses to understand the limits of transition probabilities, mortalities, costs, and utilities that could influence model outcome. These factors, together with the threshold at which they alter model outcome are shown in Table 5. The favored strategy was mainly determined by variation in the probabilities of peri-operative and annual mortality after each procedure, and the probability of annual stroke post-AVR. The thresholds of annual mortality post- AVR and post-transapical TAVI are near their reference case values noted in Table 2. Figure 4, showing a two-way sensitivity analysis comparing the impact of varying these annual mortality rates, emphasizes that slight alterations, such as a reduction of annual mortality from transapical TAVI to 20% while keeping the annual mortality post-AVR near that of the reference case, can change the outcome of the model. Likewise, Figure 5, a two-way sensitivity analysis demonstrating the interaction between the utility post- AVR and utility post-transapical TAVI, emphasizes how the utilities of each health state are also important determinants of variation in net health benefit.
| Table 5 Threshold analyses. The threshold to alter model outcome, the resultant net health benefit or net monetary benefit (at a willingness to pay of <$100,000) at that threshold, and the favored strategy are listed for all factors that modified model outcome | ||||
| Factor | Threshold value | Favored strategy (below threshold) | Net health benefit* | |
|---|---|---|---|---|
| Transition probabilities/mortality rates | ||||
| Annual mortality post-AVR | 25.53% | AVR | 1.06 | |
| Annual mortality post-transapical TAVI | 24.00% | TAVR | 1.13 | |
| Peri-operative mortality AVR | 14.53% | AVR | 1.06 | |
| Peri-operative mortality transapical TAVI | 5.05% | TAVR | 1.13 | |
| Annual stroke post AVR | 4.93% | AVR | 1.06 | |
| Cost | ||||
| Transapical TAVI | $41,904 | TAVR | $112,821 | |
| Utilities | ||||
| AVR | 0.62 | TAVR | 1.63 | |
| Transapical TAVI | 0.70 | AVR | 1.69 | |
| *Net health benefit (for willingness to pay of $100,000) is that which is obtained at the stated threshold | ||||
Variation in net monetary benefit is influenced by the initial cost attributed to transapical TAVI. The net monetary benefit from AVR exceeds transapical TAVI as long as transapical TAVI cost exceeds $41,904.
Discussion
Using existing registry data, we have shown that transapical TAVI fails to achieve incremental cost-effectiveness compared to AVR. However, with sensitivity analyses of this data, we have identified thresholds that may be helpful in identifying particular groups that would achieve greater benefit from transapical TAVI versus the population incorporated in the reference case. This model reinforces the importance of considering mortality, non-fatal adverse events, and cost in the selection between high-risk AVR and transapical TAVI.
Registry data
Given the recent development of transapical TAVI, we did not include data from registries emphasizing results of a “learning curve.” Only registries that separated recent procedures, once proper proctoring and training had been completed, were included in the data employed in the model (13). In order to ensure our analysis would compare similar groups, we also did not include registries that failed to discern between transapical TAVI performed in inoperable versus high-risk patients.
Based on the clinical features assessed in Table 1, the transapical TAVI and AVR groups were similar, except for two variables, the actual ejection fraction and the percentage of patients with peripheral arterial disease. The difference in ejection fraction is unlikely to influence outcomes, given that comparative differences in ejection fraction, when values are greater than 45%, have been shown to not influence mortality (40). Given clinical circumstance surrounding the decision to proceed with transapical TAVI, at times involving the exclusion of transfemoral access due to severe peripheral arterial disease, the high rate of peripheral arterial disease in the transapical TAVI group is concordant with current clinical practice.
Cost-effectiveness comparison with PARTNER data
This analysis focused on the comparison of outcomes between high-risk tissue AVR and transapical TAVI with the Edwards-Sapien valve. In contrast to PARTNER Cohort A, which was a randomized-control trial, we sought to study the incremental cost, health, and monetary benefit related to transapical TAVI from registries that may be more reflective of “everyday life” because of freedom from trial inclusion/exclusion criteria. In contrast to the PARTNER Cohort A cost effectiveness analysis, which assessed costs and outcomes accrued over one year post-intervention (41), a modeling approach also allows development of a longerterm perspective with the additive morbidity and mortality of each intervention assessed over a number of years. This could be important as although the patient population has a limited lifespan, the cost assessed beyond one-year postintervention could have important economic ramifications and impact the feasibility of either intervention.
The initial part of this analysis showed that the outcome of our model is consistent with previous work showing that both AVR and transapical TAVI are cost-effective when compared to standard medical management in high-risk patients, based on the superior utility derived from either strategy (42). Importantly, this confirmed the published costeffectiveness data from PARTNER, but using separate data to inform the medical management cohort, derived from a study where patients opted for medical management (25). The second component showed TAVI to be less costeffective than AVR, under our assumed scenario. The cost-effectiveness analysis from PARTNER Cohort A data showed the change in utility with the transapical approach was -0.07 QALYs; we show a similar decrement in QALYs. However, our threshold analysis (detailed in Table 5) highlights factors, that when altered from reference case values, may produce a model outcome more favorable to transapical TAVI.
Costs in our decision-analytic model were based on reimbursement determined by the weighted 2010 national estimates from the Healthcare Cost and Utilization Project (HCUP) nationwide inpatient sample. Given uncertainties in the future reimbursement and thus the actual cost of TAVI, the impact of the initial cost of TAVI, which does influence model outcome, was subject to sensitivity analysis. These results are heavily dependent on the assumption of equal reimbursement for transapical TAVI and AVR (39), and we would expect AVR to remain dominant unless lower costs pertained to transapical TAVI – perhaps this might be attainable with a shorter length of stay.
Assumptions and limitations
The modeling process is based upon assumptions regarding transition probabilities, outcomes and costs, which are informed by literature and clinical experience. Bleeding and vascular access complications overlap in various trials and registries, especially since the transapical approach itself may many times incur both types of complications in the same procedure. Accordingly, they were assessed as a single complication. Reassuringly, threshold analysis did not show the probability of transapical TAVI access site complications to have an important influence on the outcome of the model. Other post-AVR and post-transapical TAVI complications were analyzed as a single entity. The assessment of outcomes derived from weighted averages is a simplification of a variety of complications of varying severity. However, the low frequency of severe complications within an already low incidence of post-AVR complications renders this of limited influence over health outcomes (27). Similarly, in relation to transition probabilities, costs, and utilities, other less morbid conditions, such as subsequent atrial fibrillation, renal failure, or need for pacemaker placement after AVR or transapical TAVI were considered to be part of the costs and utility of the post-operative state and not included separately in our model. An analysis of the registry data employed showed no significant difference in the need for permanent pacemaker placement post-operatively (6.94% post-AVR versus 6.41% post-transapical TAVI; P=0.714), as well as the occurrence of renal failure (8.97% post-AVR versus 5.16% post-transapical TAVI; P=0.219). Given there is no comparison of these outcomes to those of patients with severe aortic stenosis receiving medical management, it is unclear how these factors would impact cost-effectiveness of transapical TAVI or AVR in this population.
Patient-prosthetic mismatch is also an important determinant of morbidity and mortality post-AVR and has also been shown to predict adverse outcomes posttransapical TAVI (43. Similarly, paravalvular aortic insufficiency has also been linked to post-operative adverse events after transapical TAVI (44). Both the occurrence of these complications and their clinical impact were assumed to be incorporated in the transition probabilities and health utilities in the model, given their limited distinction in registry data.
The most appropriate strategy to define utility for patients with two disease conditions is controversial (45). Given the high annual mortality post-AVR and post-transapical TAVI, the annual mortality of patients developing heart failure and stroke were not deemed to be significantly different than those without those conditions, and this philosophy was also applied to utilities. Thus, stroke and heart failure post-AVR and post-transapical TAVI were ascribed the mortality and utility of the worst condition. No studies have detailed the risk of posttransapical TAVI heart failure, thus this was assumed to be similar to that post-AVR.
Conclusions
With a developing technique, decision-analytic models are able to provide unique perspective not easily obtained even through clinical trials. The ICER of transapical TAVI does not satisfy standard criteria for cost-effectiveness in an unselected, non randomized-controlled trial, population defined by the literature. However, this decision-analytic model identifies patient populations that may derive superior utility at an acceptable cost with transapical TAVI, relative to surgical AVR.
Acknowledgements
Disclosure: The authors declare no conflict of interest.
References
- Smith CR, Leon MB, Mack MJ, et al. Transcatheter versus surgical aortic-valve replacement in high-risk patients. N Engl J Med 2011;364:2187-98.
- Akobeng AK. Assessing the validity of clinical trials. J Pediatr Gastroenterol Nutr 2008;47:277-82.
- Weisberg HI, Hayden VC, Pontes VP. Selection criteria and generalizability within the counterfactual framework: explaining the paradox of antidepressant-induced suicidality? Clin Trials 2009;6:109-18.
- Di Eusanio M, Fortuna D, De Palma R, et al. Aortic valve replacement: results and predictors of mortality from a contemporary series of 2256 patients. J Thorac Cardiovasc Surg 2011;141:940-7.
- Gaudino M, Anselmi A, Glieca F, et al. Contemporary results for isolated aortic valve surgery. Thorac Cardiovasc Surg 2011;59:229-32.
- Grossi EA, Schwartz CF, Yu PJ, et al. High-risk aortic valve replacement: are the outcomes as bad as predicted? Ann Thorac Surg 2008;85:102-6, discussion 107.
- Kalavrouziotis D, Li D, Buth KJ, et al. The European System for Cardiac Operative Risk Evaluation (EuroSCORE) is not appropriate for withholding surgery in high-risk patients with aortic stenosis: a retrospective cohort study. J Cardiothorac Surg 2009;4:32.v
- Kesavan S, Iqbal A, Khan Y, et al. Risk profile and outcomes of aortic valve replacement in octogenarians. World J Cardiol 2011;3:359-66.
- Leontyev S, Walther T, Borger MA, et al. Aortic valve replacement in octogenarians: utility of risk stratification with EuroSCORE. Ann Thorac Surg 2009;87:1440-5.
- Wendt D, Osswald BR, Kayser K, et al. Society of Thoracic Surgeons score is superior to the EuroSCORE determining mortality in high risk patients undergoing isolated aortic valve replacement. Ann Thorac Surg 2009;88:468-74, discussion 474-5.
- Thourani VH, Ailawadi G, Szeto WY, et al. Outcomes of surgical aortic valve replacement in high-risk patients: a multiinstitutional study. Ann Thorac Surg 2011;91:49-55, discussion 55-6.
- Gilard M, Eltchaninoff H, Iung B, et al. Registry of transcatheter aortic-valve implantation in high-risk patients. N Engl J Med 2012;366:1705-5.
- Kempfert J, Rastan A, Holzhey D, et al. Transapical aortic valve implantation: analysis of risk factors and learning experience in 299 patients. Circulation 2011;124:S124-9.
- Lefèvre T, Kappetein AP, Wolner E, et al. One year followup of the multi-centre European PARTNER transcatheter heart valve study. Eur Heart J 2011;32:148-57.
- Litzler PY, Borz B, Smail H, et al. Transapical aortic valve implantation in Rouen: Four years’ experience with the Edwards transcatheter prosthesis. Arch Cardiovasc Dis 2012;105:141-5.
- Unbehaun A, Pasic M, Drews T, et al. Analysis of survival in 300 high-risk patients up to 2.5 years after transapical aortic valve implantation. Ann Thorac Surg 2011;92:1315-23.
- Thomas M, Schymik G, Walther T, et al. Oneyear outcomes of cohort 1 in the Edwards SAPIEN Aortic Bioprosthesis European Outcome (SOURCE) registry: the European registry of transcatheter aortic valve implantation using the Edwards SAPIEN valve. Circulation 2011;124:425-33.
- Walther T, Kempfert J, Rastan A, et al. Transapical aortic valve implantation at 3 years. J Thorac Cardiovasc Surg 2012;143:326-31.
- Wenaweser P, Pilgrim T, Roth N, et al. Clinical outcome and predictors for adverse events after transcatheter aortic valve implantation with the use of different devices and access routes. Am Heart J 2011;161:1114-24.
- D’Onofrio A, Rubino P, Fusari M, et al. Clinical and hemodynamic outcomes of “all-comers” undergoing transapical aortic valve implantation: results from the Italian Registry of Trans-Apical Aortic Valve Implantation (I-TA). J Thorac Cardiovasc Surg 2011;142:768-75.
- Bosmans JM, Kefer J, De Bruyne B, et al. Procedural, 30-day and one year outcome following CoreValve or Edwards transcatheter aortic valve implantation: results of the Belgian national registry. Interact Cardiovasc Thorac Surg 2011;12:762-7.
- Eltchaninoff H, Prat A, Gilard M, et al. Transcatheter aortic valve implantation: early results of the FRANCE (FRench Aortic National CoreValve and Edwards) registry. Eur Heart J 2011;32:191-7.
- Ewe SH, Delgado V, Ng AC, et al. Outcomes after transcatheter aortic valve implantation: transfemoral versus transapical approach. Ann Thorac Surg 2011;92:1244-51.
- Sonnenberg FA, Beck JR. Markov models in medical decision making: a practical guide. Med Decis Making 1993;13:322-38.
- Wenaweser P, Pilgrim T, Kadner A, et al. Clinical outcomes of patients with severe aortic stenosis at increased surgical risk according to treatment modality. J Am Coll Cardiol 2011;58:2151-62.
- Vánky FB, Håkanson E, Svedjeholm R. Long-term consequences of postoperative heart failure after surgery for aortic stenosis compared with coronary surgery. Ann Thorac Surg 2007;83:2036-43.
- Khan SS, Trento A, DeRobertis M, et al. Twenty-year comparison of tissue and mechanical valve replacement. J Thorac Cardiovasc Surg 2001;122:257-69.
- Puvimanasinghe JP, Steyerberg EW, Takkenberg JJ, et al. Prognosis after aortic valve replacement with a bioprosthesis: predictions based on meta-analysis and microsimulation. Circulation 2001;103:1535-41.
- Syme PD, Byrne AW, Chen R, et al. Community-based stroke incidence in a Scottish population: the Scottish Borders Stroke Study. Stroke 2005;36:1837-43.
- Grunkemeier GL, Li HH, Naftel DC, et al. Long-term performance of heart valve prostheses. Curr Probl Cardiol 2000;25:73-154.
- Sullivan PW, Ghushchyan V. Preference-Based EQ-5D index scores for chronic conditions in the United States. Med Decis Making 2006;26:410-420.
- Brown PP, Kugelmass AD, Cohen DJ, et al. The frequency and cost of complications associated with coronary artery bypass grafting surgery: results from the United States Medicare program. Ann Thorac Surg 2008;85:1980-6.
- Spertus J, Peterson E, Conard MW, et al. Monitoring clinical changes in patients with heart failure: a comparison of methods. Am Heart J 2005;150:707-15.
- Tengs TO, Yu M, Luistro E. Health-related quality of life after stroke a comprehensive review. Stroke 2001;32:964-72.
- Liao L, Anstrom KJ, Gottdiener JS, et al. Long-term costs and resource use in elderly participants with congestive heart failure in the Cardiovascular Health Study. Am Heart J 2007;153:245-52.
- Healthcare Cost and Utilization Project database. 2009. Available at: http://hcupnet.ahrq.gov/hcupnet.jsp. Accessed 5/1, 2012.
- Lee WC, Christensen MC, Joshi AV, et al. Long-term cost of stroke subtypes among Medicare beneficiaries. Cerebrovasc Dis 2007;23:57-65.
- Wu Y, Jin R, Gao G,et al. Cost-effectiveness of aortic valve replacement in the elderly: an introductory study. J Thorac Cardiovasc Surg 2007;133:608-13.
- Replacement CV. Transcatheter Aortic, Transapical Aortic, and Transcatheter Pulmonary 2011. Available online: http://www.cms.gov/icd9providerdiagnosticcodes/03_ meetings.asp.
- Curtis JP, Sokol SI, Wang Y, et al. The association of left ventricular ejection fraction, mortality, and cause of death in stable outpatients with heart failure. J Am Coll Cardiol 2003;42:736-42.
- Cost Effectiveness of Transcatheter Aortic Valve Replacement Compared with Surgical Aortic Valve Replacement in Patients with Severe Aortic Stenosis; Results from The PARTNER Trial (Cohort A). Transcatheter Cardiovascular Therapeutics; 2011.
- Reynolds MR, Magnuson EA, Lei Y, et al. Health- Related Quality of Life After Transcatheter Aortic Valve Replacement in Inoperable Patients With Severe Aortic Stenosis. Circulation 2011;124:1964-72.
- Kukucka M, Pasic M, Dreysse S, et al. Patient-prosthesis mismatch after transapical aortic valve implantation: Incidence and impact on survival. J Thorac Cardiovasc Surg 2012. [Epub ahead of print].
- Sponga S, Perron J, Dagenais F, et al. Impact of residual regurgitation after aortic valve replacement. Eur J Cardiothorac Surg 2012. [Epub ahead of print].
- Fu AZ, Kattan MW. Utilities should not be multiplied: evidence from the preference-based scores in the United States. Med Care 2008;46:984-90.





