Minimally invasive valve sparing aortic root replacement (David procedure) is safe
Introduction
Minimally invasive cardiac surgery offers potential benefits of reduced surgical trauma and pain. Thus, it is gaining broader clinical application, especially in mitral valve surgery (1-3).
Composite replacement with a valved conduit, as first described by Bentall and De Bono, has been the ‘gold standard’ for the treatment of a combined pathology of the ascending aorta and the aortic valve (4). In recent years, aortic valve-sparing aortic root operations such as the re-implantation (David) procedure have become popular (5). However, valve-sparing aortic root replacements are complex procedures and demand a high level of surgical skill. Due to this reason, minimally invasive aortic root replacements are not routinely performed.
The purpose of this study was to assess the peri-operative results of minimally invasive ‘David’ procedures.
Methods
After gaining experience with more than 500 David procedures via full sternotomy, as well as more than 200 minimally invasive aortic valve replacements (AVR), a pilot project was initiated for minimally invasive ‘David Procedure’ in carefully selected elective patients.
We started with relatively young patients (<60 years) who had isolated aortic root aneurysms and aortic valve insufficiency without leaflet calcification, and no significant co-morbidities.
Accordingly, between April 2011 and March 2014, 26 patients underwent elective, minimally invasive David procedures via upper mini-sternotomy (group A). During the same time period, 14 patients (ten males, age 64.2±9.5 years) underwent elective David procedures via median full sternotomy (group B).
The pre-operative data are given in Table 1.
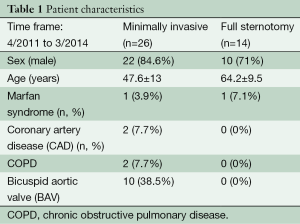
Full table
In addition to routine pre-operative examinations, coronary angiography (in patients above 40 years of age), echocardiography and computer tomography scans were performed. A David procedure was considered if the pre-operative echocardiography showed undamaged aortic valve leaflets, free of calcification. Nevertheless, the final decision to proceed with a valve-sparing operation was taken by the surgeon intra-operatively, after inspection of the aortic valve.
In group A, 14 patients (53.8%) received a Valsalva graft while 12 patients (46.2%) received a straight tube graft (David I).
Surgical procedure for minimally invasive David procedure
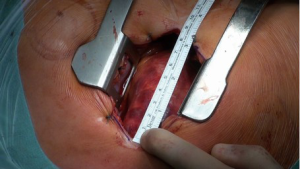
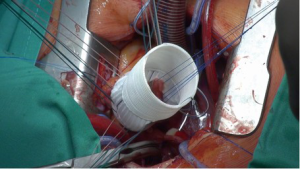
The ascending aorta and the aortic root are exposed via an upper J mini-sternotomy (up to the 3rd intercostal space) (Figure 1). When first adopting this procedure and in patients with very large aneurysms (>6 cm), it may be advisable to do the upper hemi-sternotomy up to the 4th intercostal space.
The innominate vein is identified and carefully mobilised. The pericardium is opened and the aorta visualized.
After systemic heparinization, the ascending aorta and the right atrium are cannulated directly via the mini-sternotomy access and the patient is put on cardio-pulmonary bypass (CPB). In the early learning phase, if the ascending aorta was quite large in diameter, pushing the right atrium down, venous access was performed via the femoral vein. Depending upon the extent of surgery, the patient is cooled either to 32 °C in isolated David procedures or 25 °C in case of additional aortic arch replacements.
A mediastinal chest tube and temporary epicardial pacing wires are placed via a small sub-xiphoidal incision. A CO2 sufflation line is placed into the pericardium via the mediastinal chest tube. A vent is placed into the left atrium via the upper right pulmonary vein after fibrillating the heart.
The aorta is cross-clamped and opened. Cardioplegia is given selectively through both coronary ostia. Cold blood cardioplegia (Buckberg) is our preferred method of myocardial protection during David procedures. Cardioplegia is repeated every 30 minutes.
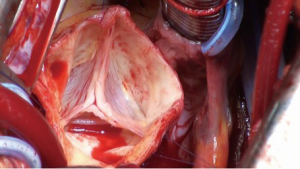
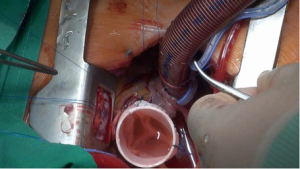
The ascending aorta is transected directly above the commissures and the aortic valve is assessed (Figure 2). The aortic root is mobilized from outside to a level immediately below the nadir of the aortic annulus. Small vessels are meticulously cauterized during aortic root preparation. Care is taken to ensure absolute hemostasis at every step of the operation.
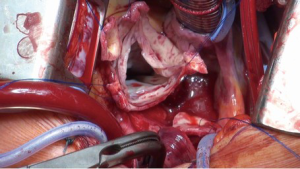
The aortic sinuses are resected to leave a rim of approximately 5 mm of the aortic wall and the coronary ostia are excised as buttons (Figure 3). If necessary, leaflet repair is performed to optimize the cusp coaptation.
The diameter of the aortic annulus is determined with a Hegar’s dilator. The diameter of the prosthesis is then calculated. The diameter of the Hegar’s dilator +2 sizes bigger determines graft diameter. In most of the patients however, the diameter of the Dacron prosthesis is either 28 or 30 mm.
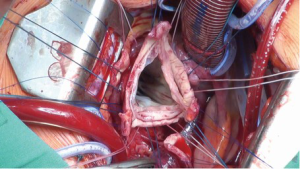
Thereafter, 9-12 unpledgeted threads of 2-0 coated polyester fiber (Ethibond, Ethicon Inc., USA) are placed, inside-out and horizontally, below the valve in a circumferential fashion (Figure 4). The Dacron graft (Gelweave or Valsalva graft, Vascutek Inc., Glasgow, Scotland) is anchored with these sutures with the aortic root inside the graft (Figure 5). The Dacron graft is fixed by tying these threads loosely to avoid the creation of a subvalvular stenosis.
If a straight tube graft is being used, the commissures are maximally pulled-up without stretching the Dacron graft and then fixed to the Dacron graft. If a Valsalva graft is used, the commissures are reimplanted at the level of the ‘neo ST junction’. The mobilised aortic root with remnants of the aortic sinuses are sutured to the inside of the Dacron graft using three 4-0 polypropylene sutures (Prolene, Ethicon Inc., USA). This is the ‘hemostatic’ suture-line and as such, has to be absolutely ‘blood-tight’.
A ‘water-test’ is performed to test the coaptation of the reimplanted aortic valve (Figure 6). Additional aortic valve leaflet repair is performed if necessary.
The coronary ostia are reimplanted to their respective neo-sinuses by using 5-0 polypropylene suture (Prolene, Ethicon Inc.). Hemostasis of the coronary anastomoses and performance of the aortic valve is tested by pressurizing the aortic root with cardioplegia. Glue is not routinely used, except in cases of calcified ostia. For this particular scenario, fibrin or Bioglue may be utilized following ‘re-implantation’ of the ostia.
The distal aortic anastomosis is then performed, and after de-airing the left ventricle, the aortic clamp is removed.
The surgical result is assessed by intra-operative transoesophageal echocardiography. After weaning the patient from CPB, meticulous hemostasis is performed before closing the chest.
Transthoracic echocardiography is again performed before discharge. Patients are anticoagulated with coumadin or aspirin (at the discretion of the individual surgeon) to prevent thromboembolic complications for only two months. Thereafter, anticoagulation therapy is discontinued unless other indications exist.
Statistical analysis
All data analyses were performed with SPSS 21.0 for Windows (SPSS Inc., Chicago, IL, USA). Normally distributed continuous variables are presented as mean ± standard deviation (SD). Numbers are given as total and relative number. Because of the small patient cohorts in both groups, no direct statistical comparison was performed.
Results
Minimally invasive David patients
There were no intra-operative conversions to full sternotomy. The cross-clamp and cardio-pulmonary bypass (CPB) times were 115.6±30.3 and 175.8±41.9 minutes, respectively. Seventeen patients (65%) could be operated without the need for any blood transfusions. The postoperative control echocardiography showed aortic valve insufficiency grade 0 in 21 patients (80.7%) and grade 1 in (19.2%) in the remaining patients. One patient was re-opened (via same access) due to post-operative bleeding. The intra-operative data are shown in Table 2.
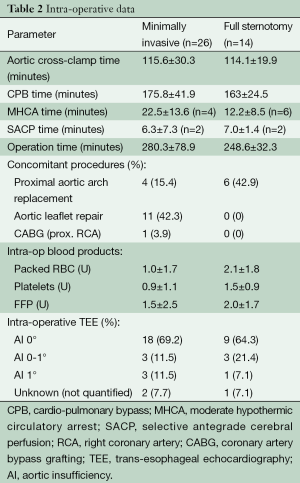
Full table
No difference in post-operative valve function was observed regardless of whether a Valsalva graft or a straight tube-graft was used.
The post-operative ventilation time and hospital stay were 0.5±0.3 and 10.4±6.8 days, respectively.
There were no deaths within the 30-day postoperative period (30 POD). The post-operative data are shown in Table 3.
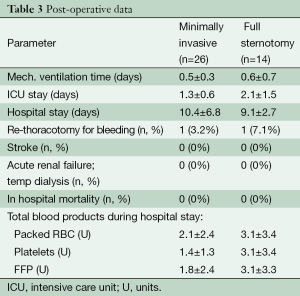
Full table
In follow-up echocardiography (477.2±321.1 days), 57.7% (n=15) patients had aortic valve insufficiency either 0° or minimal. Another 26.9% (n=7) had aortic valve insufficiency 1°. Only 15.4% (n=4) patients had aortic valve insufficiency grade 1-2. The follow-up data are presented in Table 4.
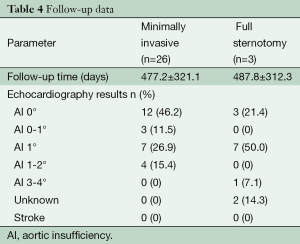
Full table
Full sternotomy David patients
The cross-clamp and CPB times were 114.1±19.9 and 163±24.5 minutes, respectively. The postoperative control echocardiography showed aortic valve insufficiency grade less than 0-1° in twelve patients (85.7%). There were no deaths within the 30-day postoperative period (30 POD). In follow-up echocardiography, one patient (12.5%) had aortic valve insufficiency either 0° or minimal. Another 62.5% (n=5) had aortic valve insufficiency 1°.
Discussion
Several studies have shown that minimally invasive AVR patients have a shorter length of hospital stay, less pain, shorter duration of ventilation, less blood loss and consequently, less blood transfusion compared to full sternotomy patients (1-3). Avoiding a full sternotomy should contribute to improved postoperative stability of the sternum and less pain. It may also reduce deep sternal wound infection. Postoperatively, the patients can be mobilized earlier and respiratory function may also normalize earlier. On the downside, limited exposure of the operative field is a disadvantage for the surgeon.
The ‘Bentall’ operation and its modifications has been seen as the ‘gold standard’ for the treatment of combined pathology of the ascending aorta and the aortic valve (4). Since the last two decades, valve-sparing aortic operations such as re-implantation (David) procedures have been proposed as alternatives (5-9). However, both of these operations are technically complex procedures. Therefore, such operations have not been done routinely via a minimally access technique.
Traversing the “learning-curve” of this approach can be made easier using a ‘step-by-step’ method. The surgeon initially garners experience by performing simpler operations such as AVR, via a minimally access. Only after a large experience (>100 such operations), the surgeon should move on to the next step of minimally access ascending aortic replacements with or without AVR. The third step should be progressing to more complex operations including Bentall procedures (aortic root replacement along with the aortic valve and re-implantation of coronary ostia). Only after gaining enough experience, we moved to minimally access valve sparing aortic root replacement (David procedure).
This ‘step-by-step’ method of progressing gradually from simpler operations to technically more complex aortic root surgery is crucial to maintaining low complication rates. Paying meticulous attention to hemostatis is a critical factor during minimal invasive access valve sparing aortic root surgery.
In our experience, the contraindications for minimally invasive David procedure were ‘Re-do’ operations and those needing concomitant cardiac procedures, such as coronary bypass operation and valve operations.
Conversion to full sternotomy was not required in any case. All the patients left the operation theatre with aortic valve insufficiency grade 0. In addition, the 30-day mortality was 0%. We used both the David I technique with a straight Dacron graft as well as a Valsalva Dacron graft. We did not find any difference between these grafts in terms of intra-operative technical ease of implantation or post-operative valve patency. The peri-operative results of these patients were comparable with those of the patients undergoing elective isolated ‘David procedures’ via full sternotomy in the same time period. However, it must be noted that the main limitation of this study is the small patient numbers in both cohorts.
In view of the fact that most the patients with a connective tissue disorder (e.g., Marfan syndrome) or those with bicuspid aortic valves present at a relative young age, it may be advantageous for the surgeons to be able to offer minimally access surgery as cosmesis is considered an important factor by these patients.
Conclusions
This pilot project shows the feasibility and safety of minimally invasive valve sparing aortic root replacements in carefully selected patients. Combining the advantages of minimally access along with that of valve sparing surgery allows the patients to return to their normal lives earlier and also does not require long-term anti-coagulation. Further experience with a larger patient cohort is needed to determine the potential clinical benefits of minimally invasive surgery for these technically complex procedures.
Acknowledgements
Disclosure: The authors declare no conflict of interest.
References
- Mihaljevic T, Cohn LH, Unic D, et al. One thousand minimally invasive valve operations: early and late results. Ann Surg 2004;240:529-34; discussion 534. [PubMed]
- Bonacchi M, Prifti E, Giunti G, et al. Does ministernotomy improve postoperative outcome in aortic valve operation? A prospective randomized study. Ann Thorac Surg 2002;73:460-5; discussion 465-6. [PubMed]
- Bakir I, Casselman FP, Wellens F, et al. Minimally invasive versus standard approach aortic valve replacement: a study in 506 patients. Ann Thorac Surg 2006;81:1599-604. [PubMed]
- Bentall H, De Bono A. A technique for complete replacement of the ascending aorta. Thorax 1968;23:338-9. [PubMed]
- David TE, Feindel CM. An aortic valve-sparing operation for patients with aortic incompetence and aneurysm of the ascending aorta. J Thorac Cardiovasc Surg 1992;103:617-21; discussion 622. [PubMed]
- Pethig K, Milz A, Hagl C, et al. Aortic valve reimplantation in ascending aortic aneurysm: risk factors for early valve failure. Ann Thorac Surg 2002;73:29-33. [PubMed]
- Leyh RG, Kallenbach K, Karck M, et al. Impact of preoperative aortic root diameter on long-term aortic valve function after valve sparing aortic root reimplantation. Circulation 2003;108 Suppl 1:II285-90. [PubMed]
- Kallenbach K, Karck M, Pak D, et al. Decade of aortic valve sparing reimplantation: are we pushing the limits too far? Circulation 2005;112:I253-9. [PubMed]
- Shrestha M, Baraki H, Maeding I, et al. Long-term results after aortic valve-sparing operation (David I). Eur J Cardiothorac Surg 2012;41:56-61; discussion 61-2. [PubMed]