Thoracoscopic diaphragm plication
Clinical vignette
A 54-year-old man presented with a history of dyspnea on exertion. His chest X-ray showed an elevated right hemidiaphragm. Diaphragm fluoroscopy demonstrated paradoxical movement. He was referred for diaphragm plication.
Surgical technique
Preparation
General anesthesia is induced using a dual lumen endotracheal tube. The patient is turned to the lateral decubitus position. We use two standard incisions: a 2-3 cm utility incision located in the anterior axillary line in the seventh intercostal space and a 5-mm camera port incision in the eighth intercostal space located anterior to utility incision (Figure 1).
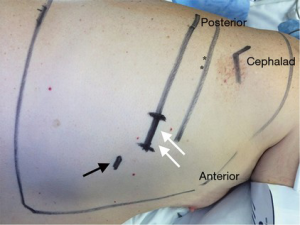
Exposition
We use a 5-mm, 30 degree angled camera for vision. After entering the thoracic cavity, exploration is performed to confirm the elevated diaphragm with thinned appearance and to ensure there are no unexpected abnormalities (Figure 2) (1).

Operation
The diaphragm is grasped, and as much of the anterior diaphragm as possible is pulled into a retaining clamp. This maneuver is easier to perform for the right diaphragm than the left diaphragm, owing to the risk of incorporating viscera on the left. Upon initial placement of the clamp, it is evident that all of the diaphragm that would optimally be resected cannot occur in one application. This is precluded by three factors: the dome of the diaphragm, the length of the staple line, and the amount of diaphragm that would optimally be resected. Rather, the clamp is progressively advanced on the redundant diaphragm after each firing of the stapler resection is progressively regrasped after each staple load. A linear endostapling device (Covidien Endo GIA™ and 45-mm Purple Reload with Tri-Staple™) is placed under the clamp, and the diaphragm resection is begun at the most anterior aspect, with the intention to resect as much of the central portion of the diaphragm as possible. The diaphragm is then resected with subsequent staple loads, serially proceeding from anterior and medial on the diaphragm, diagonally in posterior and lateral direction.
Completion
After completing the resection, the remaining diaphragm is inspected and the staple line is over-sewn continuously with a 2-0 polypropylene suture. A 24 French chest tube is placed through the camera port and the incisions are closed with absorbable sutures.
The patients are generally discharged on the following days as the chest tube is removed. On follow-up, success is indicated by either the repositioning of the diaphragm caudally on radiograph and resolution or improvement in symptoms (Figures 3,4).
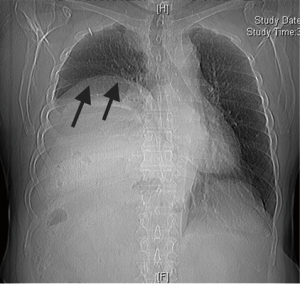
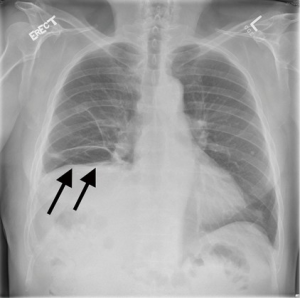
Comments
The etiology of diaphragmatic dysfunction includes both congenital and acquired conditions. True eventrations are congenital developmental defects of the muscular portion of the diaphragm having normal attachments to the ribs, sternum and dorsolumbar spine (2). Acquired diaphragmatic dysfunction is usually secondary to phrenic nerve palsy, which impairs the neuromuscular axis between spinal cord and diaphragm (2).The most important factor in the decision for surgery is the presence and severity of symptoms in the setting of demonstrated paradoxical motion of the diaphragm (2,3). The ideal treatment for symptomatic patients with diaphragmatic dysfunction is plication.
Surgical repair for diaphragm eventration was first described in 1923 and has traditionally been reserved for patients with symptoms (2). Normally, the caudal movement of the diaphragm causes the rib cage to expand producing a negative pleural pressure and causing lung inflation (2). Depending on the extent of muscle layer dysfunction and thinning the caudal movement of the diaphragm is less effective, and there is displacement of the abdominal organs cephalad into the thoracic cavity on the affected site. Both factors cause respiratory problems due to compression, reduction of the lung volumes and impaired ventilation (2). Plication increases the hemithorax volume, reduces the paradoxical motion and stiffens the diaphragm overall improving pulmonary function (2). The result is improvement in symptoms (mostly dyspnea) of the patients. Traditional lateral thoracotomy, thoracoscopy, and open or laparoscopic transabdominal approaches have been used for plication (2).
Published data about the thoracoscopic approach demonstrate plication by suturing the diaphragm either interrupted or continuous style via two to four small incisions (2,3). There have been two single case reports, one each from adult (3) and pediatric age groups (4), describing the use of endostapling devices for diaphragm plication without suture reinforcement of the staple line. Another report by Matsubara et al. describes using noncutting staplers to create two ridges, which are then sutured (5).
We describe this new technique of video-assisted thoracoscopic diaphragm plication by using endostaplers and excision of the diaphragm. We have moved our classical thoracoscopy utility incision more caudally to have better access and a better working angle on diaphragm. We utilize an endostaple device for resection of the eventrated diaphragm. We conclude the procedure with reinforcement of the stapling line by oversewing with a non-absorbable suture. One should be careful while stapling the diaphragm to ensure that there are no viscera captured within the area of excision. Securing the diaphragm with a retaining clamp as much as possible and using the clamp to guide the direction and extent of stapling greatly facilitates the resection (6,7).
Our technique is unique in that it combines the simplicity and ease of using the stapled technique with the proven security and durability of traditional hand sewn techniques. Early in our experience with thoracoscopic plication we did not oversew, but due to published and unpublished reports of staple line rupture we feel it is important to routinely oversew the staple line. Since adding the step of reinforcement by over-sewing the staple line we have performed six consecutive thoracoscopic plications and there have been no recurrences with follow up of 12 to 36 months.
This new technique for plication is safe, fast and easily applicable. Even though there is no published study comparing results of any existing interventions (2), we believe that this technique offers at least equivalent results to open plication, plus the advantages of minimally invasive surgery over classical approaches with regards to pain and recovery and promises a decrease in the operating time as compared with other minimally invasive methods.
Acknowledgements
None.
Footnote
Conflicts of Interest: The authors have no conflicts of interest to declare.
References
- Kara HV, Roach MJ, Balderson SS, et al. Thoracoscopic diaphragm plication. Asvide 2015;2:151. Available online: http://www.asvide.com/articles/728
- Groth SS, Andrade RS. Diaphragm plication for eventration or paralysis: a review of the literature. Ann Thorac Surg 2010;89:S2146-50. [PubMed]
- Moon SW, Wang YP, Kim YW, et al. Thoracoscopic plication of diaphragmatic eventration using endostaplers. Ann Thorac Surg 2000;70:299-300. [PubMed]
- Shary TM, Hebra A. A simple technique for thoracoscopic treatment of diaphragmatic eventration. Am Surg 2013;79:893-5. [PubMed]
- Matsubara H, Miyauchi Y, Ichihara T, et al. Thoracoscopic diaphragmatic plication for eventration of diaphragm in children using no-knife automatic suturing device. Kyobu Geka 2014;67:976-9. [PubMed]
- Ikeda M, Sonobe M, Bando T, et al. Reconstruction of recurrent diaphragmatic eventration with an elongated polytetrafluoroethylene sheet. Interact Cardiovasc Thorac Surg 2013;17:433-5. [PubMed]
- Takanami I. Hernia of the diaphragm with gastric ulcer and volvulus: an unusual complication after diaphragmatic resection by VATS. Interact Cardiovasc Thorac Surg 2003;2:544-6. [PubMed]





