The “UFO” procedure
Introduction
Extensive involvement of the aortic root in patients with infective endocarditis often involves the aortic-mitral curtain, also known as the intervalvular fibrous body (IVFB). It is this structure, which forms the confluence of the anterior mitral leaflet (AML), the interleaflet triangle between the left and non-coronary aortic sinuses and the roof of the left atrium. Extension of the infection into the IVFB, either due to abscess formation and/or later by development of a pseudoaneurysm, often requires complex reconstruction of the IVFB. The term “UFO” shortens the description of this complex procedure, which was coined based on a comment made by a fellow at the Toronto General Hospital who saw this complex procedure and compared it with an “Unidentified Flying Object”, something which is hard to understand, when first observed. The procedure is also known as “Combat Procedure” or the “Commando operation” in other centers.
The initial description of the basic surgical technique, which involved enlargement of the aortic root, was described by Manouguian in 1976 (1). Later in 1980, he reported a patient, who underwent aortic root enlargement, but had to be reoperated on the 4th post-operative day, as the patch tore away from the AML. He replaced the mitral and aortic valves and reconstructed the IVFB using a Dacron patch (2). The Toronto group and others later published larger series of patients undergoing this operation for a variety of pathologies such as extensive, infective endocarditis, severe aortic and mitral annular calcification and patients with small annuli requiring double valve replacement (3-7). Some of these studies also included surgical modifications like en-block replacement of the aorto-mitral curtain (6), or the use of a homograft in the aortic position (7).
It can be a useful surgical technique, not only in extensive involvement of the IVFB by infection, but also in the presence of severe calcification of this area with significant aortic and mitral annular calcification, double valve reoperations in which no IVFB is available following excision of both valves or double valve replacement in patients with extremely small aortic and mitral annuli.
The following manuscript describes the surgical steps including adequate exposure, modifications of the operation, and potential pitfalls in a step-by-step fashion.
Operative technique
Following a standard median sternotomy, cardio-pulmonary bypass (CPB) is established using bicaval cannulation. In some reoperations, initiation of CPB may be facilitated through femoral or axillary arterial and femoral cannulation. In these cases, an additional venous line for direct cannulation of the superior vena cava (SVC) or a special venous 2 stage groin cannulas enabling total CPB should be used.
The ascending aorta is cross-clamped and cardioplegia is administered directly into the aortic root or selectively into the coronary ostia, depending on the underlying aortic valve pathology.
Exposure
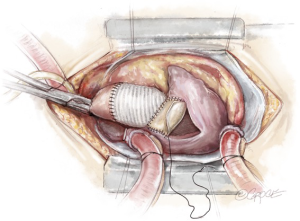
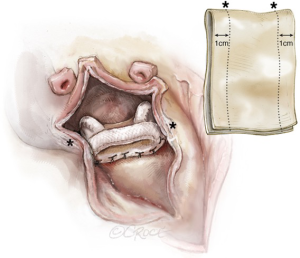
Dissection of the aortic root, especially in reoperations, can be limited or even avoided. The aorta is incised in an oblique fashion, directed towards the middle of the non-coronary sinus. In the presence of severe adhesions or extensive abscess of the aortic root, total CBP is initiated. In most cases, redo or primary, it is possible to find a plane between the aortic root and the medial wall of right atrium, so that the surgeon can dissect all the anatomical structures till the confluence of the aortic root and the left atrial roof. The aorta or the Dacron prosthesis is opened in an oblique fashion extending inferiorly up to the level of the non-coronary annulus. A separate incision is made in the left atrial roof that extends from the level of the right pulmonary artery to the base of the AML (Figure 1). However, in certain rare situations, such as large abscesses involving the roof of the left atrium and the medial wall of the right atrium, it is necessary to open the right and left atria in a modified fashion as described previously by Guiraudon et al. (8). The modification consists of an incision that starts in the lateral wall of the right atrium, extends across the right atrial appendage into the medial wall, which is then incised along with the non-coronary sinus of the aorta towards the atrial septum. The latter is partially opened (until the superior edge of the fossa ovalis) and the incision is further extended into the roof of the left atrium to reach its confluence with the AML and the aortic annulus (Figure 1).

The AML is completely excised, all macroscopically infected tissue should be radically removed. (Alternatively, all calcified tissues in patients with annular calcification should be resected). The entire IVFB is, therefore completely excised. In rare cases, in which the abscess extends into the central fibrous body towards the tricuspid valve, the right atrial incision may be extended to this area. It is usually in the direction of the anterior-septal commissure. This approach can also be used in some cases, in which a defect between the left ventricle and the right atrium exists, as first described by Buhl (9).
Thereafter, the surgeon has a very good view of the entire aortic root, the left atrial and ventricular cavities and the posterior aspect of the mitral valve. Performance of an aortic valve or total aortic root replacement depends on the extent of involvement of the aortic root. If the latter is necessary, the aortic sinuses are excised and coronary artery buttons are prepared (Figure 2). In essence, the left ventricle is now akin to a cone that has a single large opening. This is the watershed stage of the operation, when resection ends and reconstruction begins. All instruments, suction cannulas and gloves should be changed in patients with endocarditis. A thorough wash should be given.
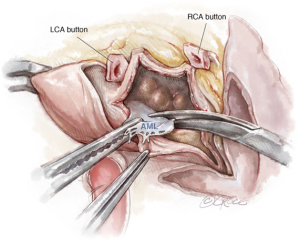
Mitral valve replacement
Reconstruction of the inlet and outlet of the left ventricle begins with mitral valve replacement. Routine mitral valve sizing is not possible. Oversizing the mitral valve prosthesis should be avoided as this may result in obstruction of the left ventricular outflow tract (LVOT) and implantation of a smaller than desired aortic valve substitute. The PML is preserved in patients, in whom it is not involved in the pathological process. Nevertheless, in the presence of severe posterior mitral annular calcification or abscess formation, complex annular reconstruction techniques can be performed, as the visualization on the posterior mitral annulus is excellent.
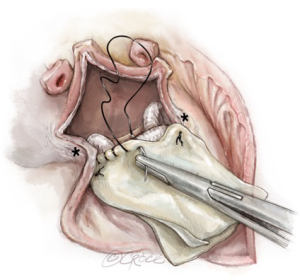
Pledgeted sutures are placed through the posterior aspect of the mitral valve annulus, from the lateral to the medial fibrous trigones (Figure 3).
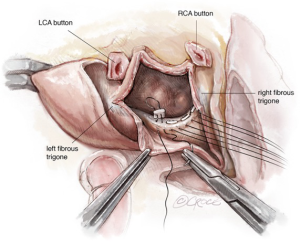
Sutures are passed through the corresponding part of the sewing cuff of the selected mitral valve prosthesis. Attention to accurate valve positioning is important as it is possible to lose orientation, when the usual landmarks do not exist. The hinge mechanism should be aligned along the antero-posterior diameter in case of mechanical valve prostheses and the 2 anterior struts of the bioprostheses should be on either side of the LVOT (at the 2 fibrous trigones). Positioning of the struts in the LVOT should be avoided (Figure 4).
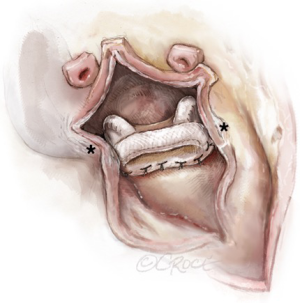
Reconstruction of the left atrial roof
Reconstruction of the left atrial roof and, if necessary, part of the interatrial septum is accomplished by using either a two- or single-patch technique. In the two-patch technique, a large bovine pericardial patch is folded on itself and is trimmed so that the distance between the lateral and medial fibrous trigones correspond to the width of the patch, which is sutured to the anterior sewing cuff of the mitral valve prosthesis starting from each of the fibrous trigones (Figure 5). It is very important to confirm that the 2 points of transition (the 2 trigones) from the native posterior annulus to the anterior neo-pericardial annulus are water-tight as bleeding, paravalvular leakage or shunting can easily occur at these points, especially in the presence of delicate or fragile tissue (Figure 6). One can further reinforce the patch at both trigones with reinforcing sutures, however, the best technique of achieving this is to pass the valve sutures at the 2 native trigones through the 2 ends of the folded pericardial patch before passing them through the sewing cuff of the mitral prosthesis. This results in the pericardial patch being sandwiched between the native annulus and the sewing cuff when the mitral prosthesis is lowered down and tied. The posterior tongue of the patch is used to reconstruct the roof of the left atrium. Visualization can be enhanced by invaginating the anterior tongue into the LVOT. The anterior tongue is later used to reconstruct the non-coronary sinus of the aortic root or, is trimmed and used to anchor the aortic conduit (Figure 7).
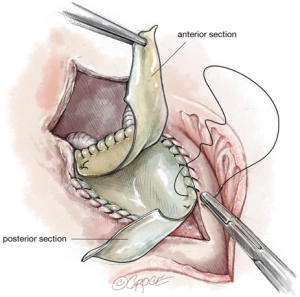
If the infection progresses towards the tricuspid valve, it is advisable to reconstruct the resected parts of the right atrium with an additional pericardial patch before the mitral valve is replaced.
Alternatively, a single patch can be used with further trimming of the part, which is sutured to the anterior aspect of the mitral valve prosthesis (Figure 8).
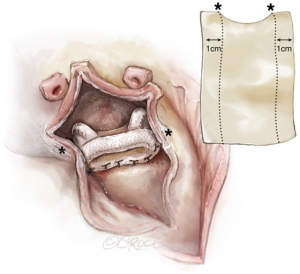
The patch can also be used, if part of the atrial septum has to be replaced. Care has to be taken to measure the adequate length of the patch, to avoid tilting of the prosthesis into the LVOT. The single patch technique has the advantage of improved visibility of the operative field.
Aortic root procedure
There are different options available for reconstruction of the aortic root and aortic valve replacement. If only the non-coronary sinus requires resection, it can be reconstructed by suturing the anterior tongue of the pericardial patch to the anterior and posterior margins of the native aorta and aortic root, before the aortic valve prosthesis is implanted. In the single patch technique, a second patch is sutured to the anterior aspect of the mitral valve.
If, however, an aortic root replacement with a valve conduit is required, the anterior part of the patch is sutured for a distance of about 1.5 cm to the anterior and posterior margins of the LVOT and is then trimmed. This forms the new IVFB, the upper edge of which provides an adequate rim for anchoring the aortic valve conduit. This step is especially important to maintain adequate distance between the aortic and mitral mechanical valve prostheses in order to avoid interference in their leaflet motion.
In patients with severe calcification or extensive endocarditis, radical resection of the aortic root and IVFB often renders proper anchoring of the aortic valve conduit in the LVOT challenging. In such cases, the native aortic valve annulus is completely destroyed, and the conduit has to be anchored deep into the LVOT. This can be accomplished by tailoring a custom-made aortic valve conduit (mechanical or biological) such that a 1–1.5 cm skirt of a Dacron prosthesis remains under the aortic valve prosthesis (Figure 9). To accomplish this, the aortic valve prosthesis is either introduced into the Dacron graft and sutured from outside or the graft is folded on itself for approximately 1–1.5 cm, after which the aortic valve prosthesis is introduced into the Dacron graft and sutured to its folded margin. Thereafter, the folded part of the graft is straightened again by bringing it over the prosthetic valve, which is thus positioned within the graft with a 1–1.5 cm skirt under it (Figure 10). The Dacron graft should be at least 5 mm larger than the size of the aortic valve prosthesis.
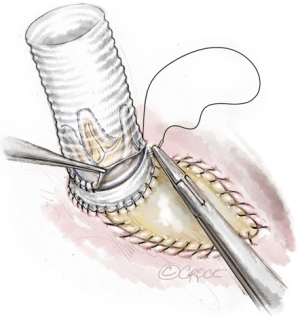
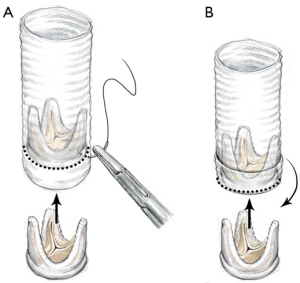
An alternative way to reconstruct the aortic root is to directly suture the Dacron graft to the LVOT. The technique used can either be the same as that for an aortic valve replacement, which is performed by using interrupted horizontal mattress pledgeted polyester sutures passed through the LVOT and the Dacron graft, or by inverting the Dacron graft into the left ventricle (Figure 11A) and suturing it to the LVOT with a continuous polypropylene suture in 1 or 2 layers. (Figure 11B). Thereafter, the Dacron graft is pulled out of the left ventricle and the aortic valve prosthesis can then be sutured to it with the technique already described above (Figure 10A).
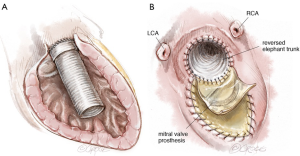
It is of utmost importance to confirm that the aortic valve conduit is securely anchored in the LVOT, as bleeding from this proximal suture line is usually not accessible and can become fatal. An important detail that needs attention is the level at which the prosthetic aortic valve has to be sutured within the graft so as not to jeopardize the reimplantation of the coronary buttons.
Rarely, the coronary buttons cannot be reimplanted directly, most commonly due to severe adhesions encountered during reoperations. In such situations, a modified Cabrol procedure may be required (10). A 6–8 mm Dacron graft is anastomosed to each coronary ostium. Following completion of the distal aortic anastomosis, each of the coronary Dacron grafts is sutured to the aortic valve conduit, taking care to avoid any kind of kinking (Figure 12).

Reconstruction of the right atrium
In patients in whom the Guiraudon incision is used, the right atrium is closed with an additional pericardial patch, if necessary, following reconstruction of the aortic root, (Figure 13).
Dealing with complications
The “UFO” procedure is a very complex surgical technique with a high risk of intra-operative complications. This is especially true in patients with infective endocarditis with fragile tissues, which are prone to tear or crumble leading to life-threatening bleeding, paravalvular leaks or rupture, after the cross-clamp has been released and the root pressurized. The trigonal areas, to which the single or double pericardial patch is attached, are at the highest risk for such complications Paravalvular leaks are usually detected in the area of the mitral valve prosthesis and the reconstructed IVFB by intra-operative transesophageal echocardiography. The surgeon has to balance the risk of reopening at least the roof of the left atrium resulting in a partial “take down” of the reconstruction versus the potential adverse hemodynamic effects of refraining from surgically addressing the leak. This obviously depends on the severity and site of the leak. If anatomically suitable, interventional procedures, using occluder systems, may be considered in the postoperative period.
In case of uncontrolled bleeding from the root, a Cabrol patch or its modifications may be used to tide over the crisis (10). If the bleeding is not severe, this strategy is often helpful, especially in reoperations, in which the transverse sinus is closed under the pulmonary artery due to adhesions. In the event of more severe bleeding, a communication may be established between the extracardiac chamber developed by the Cabrol patch and the right atrium in order to decompress it. Significant ruptures are not well tolerated as they have to be immediately repaired and that increases the ischemic and CPB times to levels that may be life-threatening.
Comments
The “UFO” procedure is not a medical term. However, it shortens the description of a very complex surgical repair, involving the aortic and mitral valves, the IVFB and in some cases even other adjoining cardiac structures. In patients with infective endocarditis, adopting the surgical strategy of radical resection of all infected and prosthetic tissue followed by complex reconstruction of all excised cardiac structures and tissue with the use of bovine pericardium is required. A meticulous technique, a thorough knowledge of anatomy, a creative 3D imagination of the heart, surgical talent and the ability to innovate “ad hoc” are all required to get maximum possible success from these operations.
Although the overall operative morbidity and mortality is of this procedure is high (4,5,11), it continues to remain the only option for patients presenting with destroyed or heavily calcified aortic and mitral annuli.
Acknowledgments
The authors would like to thank Beth Croce for creating the brilliant images.
Footnote
Conflicts of Interest: The authors have no conflicts of interest to declare.
References
- Manouguian S. Erweiterungsplastik des hypo-plastischen Aortenklappenringes: Beschreibung einer neuen Operationsmethode: eine experimentelle Studie. Thorac Cardiov Surg 1976;24:418-23. [Crossref]
- Manouguian S, Kirchhoff PG. Patch enlargement of the aortic and the mitral valve rings with aortic-mitral double-valve replacement. Ann Thorac Surg 1980;30:396-9. [Crossref] [PubMed]
- David TE, Kuo J, Armstrong S. Aortic and mitral valve replacement with reconstruction of the intervalvular fibrous body. J Thorac Cardiovasc Surg 1997;114:766-71; discussion 771-2. [Crossref] [PubMed]
- Davierwala PM, Binner C, Subramanian S, et al. Double valve replacement and reconstruction of the intervalvular fibrous body in patients with active infective endocarditis. Eur J Cardiothorac Surg 2014;45:146-52. [Crossref] [PubMed]
- Forteza A, Centeno J, Ospina V, et al. Outcomes in aortic and mitral valve replacement with intervalvular fibrous body reconstruction. Ann Thorac Surg 2015;99:838-45. [Crossref] [PubMed]
- Hassan M, Windsor J, Ricci M. En bloc aortic and mitral valve replacement and left ventricular outflow tract enlargement using a combined transaortic and trans-septal atrial approach. Interact Cardiovasc Thorac Surg 2015;21:792-5. [Crossref] [PubMed]
- Hosseini MT, Kourliouros A, Sarsam M. Double-valve endocarditis homograft and patch repair. Ann Thorac Surg 2009;88:1708-9. [Crossref] [PubMed]
- Guiraudon GM, Ofiesh JG, Kaushik R. Extended vertical transatrial septal approach to the mitral valve. Ann Thorac Surg 1991;52:1058-60; discussion 1060-2. [Crossref] [PubMed]
- Meyer H. Uber angeborene Enge oder Verschluss der Lungenarterienbahn. Virchow's Arch f Path Anat 1857;12:532. [Crossref]
- Cabrol C, Pavie A, Gandjbakhch I, et al. Complete replacement of the ascending aorta with reimplantation of the coronary arteries: new surgical approach. J Thorac Cardiovasc Surg 1981;81:309-15. [PubMed]
- De Oliveira NC, David TE, Armstrong S, et al. Aortic and mitral valve replacement with reconstruction of the intervalvular fibrous body: an analysis of clinical outcomes. J Thorac Cardiovasc Surg 2005;129:286-90. [Crossref] [PubMed]