Remodeling of the aortic root—a 28-year journey
Introduction
Root remodeling was designed as an alternative to combined valve and root replacement as treatment for aortic regurgitation (AR) and root aneurysm (1). According to the original hypothesis that AR was due to aortic dilatation, normalizing root dimensions should lead to normal aortic valve function. While early results reported good outcomes (2), late results of the original series showed that a relevant proportion of patients required reoperation for recurrent AR (2). This was assumed to be related to the lack of annular stabilization (3,4), questioning the long-term value of the technique.
We started to explore the role of root remodeling 28 years ago (5). We encountered the obvious occurrence of concomitant cusp prolapse in the presence of root aneurysm, which we treated by concomitant cusp repair with root remodeling (6). The addition of cusp repair did not lead to inferior short- and mid-term results (7). In parallel to the clinical application, we investigated cusp motion in an in-vitro study and compared it to aortic valve reimplantation (8). Interestingly, cusp motion was more physiologic with root remodeling compared to valve reimplantation (8), which encouraged us to continue employing the concept. We modified the procedure to accommodate the characteristics of the bicuspid (9), and later, the unicuspid aortic valve (UAV) (10).
Initial valve assessment relied on visual inspection only. The analysis of failed bicuspid aortic valves (BAV) led to the recognition of normal aortic valve configuration. We developed the concept of effective height (eH) (cusp margin in diastole to annular plane) as a cusp configuration parameter (11,12). We also defined the amount of cusp tissue of normal aortic valves—termed geometric height (gH) (11). Analyzing early and mid-term results, we found equivalent valve function with root remodeling and valve reimplantation (13). Stimulated by the need for an annuloplasty in isolated BAV repair, we subsequently applied a suture annuloplasty in remodeling (14). Thus, in the past 18 years root remodeling has been a standardized procedure based on geometric principles.
The objective of the analysis was to review the long-term results of root remodeling into the third postoperative decade.
Methods
Patients
We conducted a retrospective analysis of 1,189 patients (Table 1) who underwent root remodeling at Saarland University Medical Center between October 1995 and September 2022. The investigation was approved by the Saarland Regional Ethics Committee (CEP 202/19, CEP 203/19), and individual patient consent was waived for the analysis and publication due to its anonymized nature.
Table 1
| Characteristics | N=1,189 |
|---|---|
| Male sex, n [%] | 899 [76] |
| Age, mean ± SD, years | 53±14 |
| BSA, mean ± SD, % | 1.8±0.4 |
| Cardiovascular risk factors, n [%] | |
| Arterial hypertension | 855 [72] |
| Coronary artery disease | 133 [11] |
| Chronic kidney disease | 38 [3] |
| Chronic obstructive lung disease | 38 [3] |
| Surgical indication, n [%] | |
| Isolated aortic regurgitation | 700 [59] |
| Aortic root dilatation (≥50 mm) | 595 [50] |
| Acute aortic dissection | 84 [7] |
| Combined disease | 5 [0.4] |
| Prior aortic valve operation, n [%] | 127 [11] |
| Valve morphology, n [%] | |
| Unicuspid | 33 [2] |
| Bicuspid | 472 [40] |
| Tricuspid | 684 [58] |
| LVEF <50%, n [%] | 89 [8] |
| LVEDd, mean ± SD, mm | 56.0±7.7 |
BSA, body surface area; LVEDd, left ventricular end-diastolic diameter; LVEF, left ventricular ejection fraction; n, number; SD, standard deviation.
Surgical technique
Intraoperative transesophageal echocardiography (TEE) was performed for assessment of aortic root dimensions and cusp pathology. The surgical technique depended on the valve morphology and cusp pathology encountered, including cusp repair and suture annuloplasty as needed. The technique and its modification for bicuspid and unicuspid valves have been described in detail previously (5,10,15).
Briefly, all operations were performed via a median sternotomy with aortic and right atrial cannulation. In acute dissection, the right axillary artery was used for arterial inflow. Antegrade blood cardioplegia was given directly into the coronary ostia for myocardial protection.
Cusp size was determined before deciding in favor of valve preservation. In tricuspid valves, a gH >18 mm allowed for valve preservation. In bicuspid valves, a gH of >20 mm of the nonfused cusp was taken as trigger for valve preservation. In unicuspid valves, a gH of >20 mm measured on the pliable tissue of the left and non-coronary cusps allowed for repair.
The decision for valve replacement was made generally for cusp calcification, active endocarditis and retraction in bicuspid valves (n=103/1,168; 8.8%). Tricuspid aortic valve (TAV) was most commonly replaced for cusp retraction, multiple fenestrations or calcification (n=80/726; 11%). In UAVs, extensive calcification beyond the limits of the right cusp triggered replacement.
After root mobilization and excision of the sinus wall, a tubular graft was tailored to accommodate the configuration of the aortic root and sutured to the cusp insertion lines. The length of the tongues was adjusted according to the height of the native commissures; an effort was made to create tongues roughly 1 to 1.5 cm longer than the commissural height.
Originally, remodeling was used for patients with an annular diameter of <30 mm, and the chosen graft size was 1 to 2 mm smaller than the basal diameter. Later, all root morphologies were included, and graft size was chosen according to the body surface area of the patient (24 mm for <1.8 m2, 26 mm for 1.9 to 2.2 m2, and 28 mm for 2.3 m2 and larger). In TAVs with a gH <20 mm, a smaller graft (one size less) was taken.
Valve configuration was assessed after completing the root procedure. Initially, the valve was inspected visually (n=243; 23%). Starting in 2004, eH of each cusp was determined using a caliper (Fehling Instruments, Karlstein am Main, Germany) (n=804; 77%) (Figure 1) (11). Cusp prolapse was defined as eH <9 mm (in BAV measured in the nonfused cusp) and corrected by central plication until an eH of 9 to 10 mm was reached (TAV n=609, BAV n=336, UAV n=33). Small fenestrations were accepted if they were not involved in prolapse. Perforations and larger fenestrations were closed with a pericardial patch (autologous pericardium n=26; heterologous pericardium n=6).
An external annuloplasty has been added since 2009 if the annulus measured >26 mm. In most instances (n=524), an expanded polytetrafluoroethylene suture (Gore-TexCV-0, W. L. Gore & Associates, Munich, Germany) was used (Table 2). The suture was tied around a Hegar dilator (<1.8 m2: 21 mm, 1.8 to 2.0 m2: 23 mm, >2.0 m2: 25 mm). The Hegar size was reduced by 2mm in tricuspid valves with gH <19 mm and bicuspid valves with gH <22 mm.
Table 2
| Variables | N=1,189 |
|---|---|
| Annular support, n [%] | |
| Polytetrafluoroethylene suture | 528 [44] |
| Two-braided polyester | 84 [7] |
| Cusp repair, n [%] | 1,047 [88] |
| Central plication | 972 [93] |
| Measurement of effective height | 804 [77] |
| Cusp pathology, n [%] | |
| Cusp prolapse | 972 [82] |
| Fenestrations | 34 [3] |
| Retraction | 6 [0.5] |
| Perforations | 5 [0.4] |
| Patch material, n [%] | 95 [8] |
| Autologous pericardium | 78 [82] |
| Decellularized matrix patch | 15 [16] |
| Synthetic material | 2 [2] |
| Concomitant procedure, n [%] | |
| Hemi-arch using circulatory arrest | 324 [27] |
| Coronary artery bypass | 133 [11] |
| Atrial ablation | 62 [5] |
| Mitral valve repair | 40 [3] |
| Perfusion time, mean ± SD, min | 92±21 |
| Myocardial ischemia, mean ± SD, min | 68±14 |
| Perioperative complications, n [%] | |
| Bleeding | 30 [2.5] |
| Permanent pacemaker implantation | 1 [0.1] |
| Neurological complication | 2 [0.2] |
n, number; SD, standard deviation.
In bicuspid valves, triangular resection of raphe tissue was performed in the presence of dense fibrosis or limited calcification (n=129). The fused cusp was reconstructed directly (n=103) or augmented by an autologous pericardial patch tissue (n=26). For symmetric or moderately asymmetric BAV, the commissures of the non-fused cusp were placed at a 160° orientation in the first 119 patients. In all subsequent patients, an orientation of approximately 180° was chosen with two symmetric tongues for symmetric and asymmetric BAV (n=309). In very asymmetric BAVs, three tongues were created in analogy to TAVs (n=44).
In unicuspid valves, the left-noncoronary commissure is usually the functioning commissure and was used as a reference for commissural height. The new commissure was created opposite of this left-noncoronary commissure for symmetric orientation (16). Two triangular patches were prepared to bridge the gaps between preserved left or non-coronary cusp tissue and the new commissure. In six patients, cusp nadir relocation was performed without the use of a patch (17).
Concomitant procedures were performed first, followed by root remodeling (n=506; 43%), most commonly hemi-arch replacement (n=324) (Table 2).
All patients underwent intraoperative TEE. They also underwent transthoracic echocardiography (TTE) before discharge, at three months, at one year and biannually thereafter. Mean and peak systolic gradients were measured, and AR was analyzed by color Doppler and classified as absent, mild, moderate or severe.
Follow-up
All patients were seen regularly by their referring cardiologists or in our clinic. Echocardiograms from our institution and referring cardiologists were reviewed. All patients were followed prospectively both clinically and echocardiographically (at discharge, 3 months, 1 year and yearly thereafter). Systolic gradients were measured using continuous wave Doppler. AR was determined using color Doppler according to European guidelines.
Median and mean follow-up were six years [range, one month to 28 years] and 6.7±5.5 years. Follow-up was 95% complete (7,700 patient-years).
Statistical analysis
Non-normally distributed continuous variables are presented as median (interquartile range), and the Mann-Whitney U test was used for between-group comparisons. Normally distributed continuous variables are presented as mean ± standard deviation (SD) and were compared using the t-test. Categorical variables are expressed as frequency (%). Time-dependent data were analyzed using the Kaplan-Meier method. Differences were assessed using the log-rank test. Survival and freedom from reintervention were calculated at one, five, ten, 15 and 20 years. All statistical tests were two-sided, and P values <0.05 were considered statistically significant for all analyses. In logistic regression analysis, a P value <0.10 in the univariable analysis was eligible for entry into the multivariable analysis. We applied a stepwise procedure for selecting variables based on the Wald criterion of forward induction. Statistical analyses were performed using SPSS 28.0 (IBM Corp. Released 2021. IBM SPSS Statistics for Macintosh, Version 28.0. Armonk, NY: IBM Corp).
Results
Patients
Of all patients, 76% were male with a mean age of 53±14 years (range, 2 to 86 years) (Table 1). The original aortic valve morphology was unicuspid in 33 (2%), bicuspid in 472 (40%), and tricuspid in 684 (58%) patients. Fifty-four patients (5%) had connective tissue disease, in the majority of cases Marfan’s syndrome. Prior to the index procedure, 127 patients (11%) had undergone at least one cardiac operation. The primary indications for surgery were severe and symptomatic AR (n=696) and aortic root dilatation (sinus ≥50 mm, n=595). Eighty-four patients underwent remodeling for acute aortic dissection (7%; Table 1).
Early
Cusp pathology requiring correction included cusp prolapse (n=972; 82%), fenestrations (n=34), retraction (n=6) and perforations (n=5). Cusp repair was performed in 1,047 (88%) patients. A patch was used in 95 patients (8%). It was used for partial cusp replacement or augmentation in BAVs (n=37), TAVs, for closure of cusp perforations and fenestrations (n=32), and for UAV repair (n=27). Autologous pericardium was used as patch material in 78 patients (82%), heterologous pericardium in 15 (16%) and PTFE in 2 (2%; Table 2).
Mean myocardial ischemia and extracorporeal perfusion times were 85±20 and 121±35 minutes with concomitant procedures and 68±14 and 92±21 minutes, respectively, without concomitant procedures (P<0.001) (Table 2).
There was no myocardial infarction and two patients developed neurological complications. One patient required permanent pacemaker implantation after ablation for persistent atrial fibrillation; no atrioventricular block was observed in patients with sinus rhythm. There were no early valve reoperations; re-exploration for bleeding was necessary in 30 patients (2.5%; Table 2).
Hospital mortality was 1.5% (n=18/1,189). Causes of death included non-occlusive mesenteric ischemia (n=3), sepsis (n=5), cardiac failure (n=5), pulmonary embolism (n=1), cardiac arrhythmia (n=1), cardiac tamponade (n=1), cerebral hemorrhage due to an intracranial aneurysm (n=1) and stroke (n=1).
AR at discharge
With the introduction of eH measurement, the proportion of patients with only AR <1 at discharge increased (n=675; 82.4%) compared to without eH measurement (n=170; 71%).
With suture annuloplasty, a higher proportion of patients had only minimal recurrent AR at discharge (n=565/624; 91%) than without suture annuloplasty (n=412/532; 77%; P<0.001).
Late survival
Late postoperatively, 123 (10%) patients died (between 1.1 months and 23 years). Of these patients, 82 (67%) died of a cardiac cause. Survival was 71% at 20 years; freedom from cardiac death was 80% at 20 years (Figure 2) (TAV: 80%; BAV: 95%; UAV: 100%; P<0.001; Figure 3A). Survival at 15 years was significantly better in patients who underwent root remodeling as an elective operation (76%) in comparison to acute aortic dissection (58%; P<0.001; Figure 3B). Of the patients who underwent the operation as an elective procedure, those who underwent partial arch replacement had lower survival rates (68%) compared to those without partial arch replacement (79%; P=0.004). It was also superior at 15 years in patients without a concomitant coronary artery bypass graft (CABG) procedure (79%) compared to those with CABG (54%) (P<0.001).
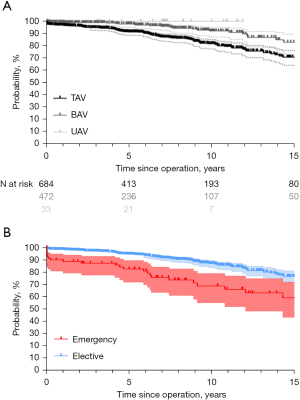
The measurement of eH improved survival at 15 years (with 78%; without 52%; P<0.001). Survival at 12 years was higher in patients with annuloplasty compared to those without (90% vs. 71%; P<0.001)
By univariable analysis, age (P<0.001), male gender (P=0.008), concomitant procedures (P<0.001) and the lack of eH measurement (P<0.001) were predictors for late death. By multivariable analysis, patient age [odds ratio (OR) =1.1] and absence of eH measurement were predictors for late death (OR =6.07).
Freedom from recurrent AR ≥2
Valve function has remained stable in the majority of patients. Over time, however, 104 patients (8%) developed AR ≥2. Freedom from AR ≥2 was 90% at 10 years and 77% at 15 years (Figure 4) (TAV 90%; BAV 88%). Freedom from AR ≥2 in UAVs was 88% at 10 years.
At 10 years, there was a trend towards a better freedom from AR ≥2 with the addition of a suture annuloplasty (92%) compared to patients without a suture annuloplasty (87%; P=0.07). Patients who underwent plication of all three cusps, however, had a lower freedom from recurrent AR ≥2 at 10 years (P<0.001) compared to those with repair of 1 or 2 cusps only.
Gradients
In patients with TAV, normal systolic gradients (mean 4±3 mmHg) remained throughout the follow up in almost all cases. With BAV, the mean gradient at last follow-up was 7±6 mmHg. It was 10±8 mmHg with asymmetric orientation compared to 6±5 mmHg when symmetric orientation was performed (P=0.03).
Reoperation
Sixty-nine patients required aortic valve reoperation between one month and 21 years postoperatively (median six years). The main indications for reoperation included recurrent AR ≥2 (n=40), active endocarditis (n=11) and aortic stenosis (BAV n=6, TAV n=3).
The main findings at reoperation were suture dehiscence (n=17), active endocarditis (n=11), persistent/recurrent cusp prolapse (n=14), retraction (n=9) and unrepaired fenestrations leading to prolapse (n=4). Reoperations consisted of valve replacement (n=37), valve repair (n=21), root replacement (n=6) and pulmonary autograft replacement (n=5).
Overall freedom from reoperation was 88% at 20 years (Figure 5). Freedom from reoperation at 15 years was superior in TAV (94%) compared to BAV (84%) and UAV (P<0.001). Freedom from reoperation for UAV was only available up to ten years (64%; Figure 6A). Patients who underwent the procedure for acute aortic dissection had a similar freedom from reoperation at 15 years (93%) compared to those who underwent an elective procedure (90%). Patients with connective tissue disease also had a similar freedom from reoperation at 15 years (97%) compared to those without (90%) (P=0.242). AR as the primary indication for surgery did not affect freedom from reoperation at 15 years (92% with AR vs. 89% without; P=0.63).
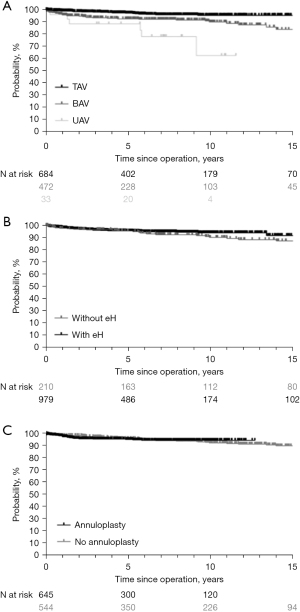
Since the introduction of eH measurement, freedom from reoperation has remained stable at 15 years and was 92% with and 87% without (Figure 6B; P=0.273). It was 92% without and 97% with eH measurement in tricuspid valves, and 83% without and 85% with eH measurement in bicuspid valves. There was also no difference between the number of cusps that were plicated (P=0.390).
Freedom from reoperation at 12 years was 95% with the addition of a suture annuloplasty and 91% without (P=0.949; Figure 6C). It was 94% without and 97% with annuloplasty in TAV, and 88% without and 92% with annuloplasty in BAV.
Discussion
Root remodeling was originally designed as an alternative to conventional valve and root replacement assuming that AR was due to aortic dilatation, and normalization of root dimensions should lead to normalization of aortic valve form and function (18). Good early results were reported (2); subsequently, however, the analysis of late results showed a relevant proportion of recurrent AR (2,4). The reasons for these failures were not clear, although the lack of annular stabilization was proposed to be a likely mechanism (3,4). While these late results questioned the principal value of this surgical technique, we were positively impressed by the more physiologic cusp motion and systolic gradients in in-vitro experiments (8,19). The lesser need for aggressive basal dissection compared to aortic valve reimplantation and the somewhat shorter ischemic time (20) prompted us to use root remodeling on a routine basis. While the operation was originally designed for the TAV anatomy, we modified the procedure to accommodate the anatomy of a BAV (15). More recently, we also applied it to UAV anatomy (10).
Almost three decades after our first use of root remodeling, the basic principle of the operation has remained unchanged. Compared to the original technique (2) only minor details were changed and then kept constant throughout our practice: the length of the Dacron tongues was not predetermined but rather adjusted to native commissural height in such a way that it exceeded the height of the native commissures. In order to facilitate the adjustment, suturing was always started in the sinus nadir and completed at the commissures. The only conceptual modifications over time have been (I) The addition of cusp repair, (II) the introduction of systematic measurement of eH to standardize detection and correction of cusp prolapse and (III) the addition of an annuloplasty (Video 1).
Over time, we observed that root aneurysm not infrequently coexisted with cusp prolapse, which was defined by visual inspection during the operation (6,21-24). Concomitant correction of prolapse by plication of redundant cusp tissue was started, and we found that aortic valve durability was not impaired (6). We later recognized that the frequency of cusp prolapse seemed to correlate with the severity of AR (25). This prolapse is often not apparent at preoperative echocardiography because the stretching of cusps is masked by root aneurysm with increased intercommissural distance (26). The correction of sinotubular dilatation by reducing the intercommissural distance can thus induce and/or aggravate cusp prolapse (26), a phenomenon that we first detected in preserved BAVs. Therefore, we started to systematically assess for possible prolapse and its correction after completion of root remodeling.
Most importantly, the analysis of failed BAVs stimulated us to investigate the normal form of an aortic valve. We hypothesized that the height difference between annular plane and cusp margins in diastole—termed eH—could be used as a configuration parameter for the aortic valve (11). In a prospective trial with volunteers and normal aortic valves, we found a close correlation between eH and sinus dimensions as well as body surface area (12), with 9 to 10 mm being ideal for normal-sized adults. This was confirmed by a later anatomical study (27). In a retrospective analysis of aortic valve repair patients (28), we also confirmed that an eH of 9 mm or more was a good predictor of valve competence and durability. In effect, we introduced the systematic measurement of eH into our assessment of aortic valves in 2004 with a caliper that allowed for intraoperative measurement. This measurement became a routine part of our valve-sparing procedures and prolapse was defined as an eH of <9 mm (11).
It was clear from the beginning that eH also had to take into consideration the amount of cusp tissue (11). As simple surrogate marker of cusp size, we used the distance from the nadir to the center of the free margin, termed gH. In the absence of plausible data, we initiated a prospective clinical trial in which we measured gH in BAVs and TAVs (29). The mean gH for TAV was 20 mm. This was confirmed by a later anatomical study (27,30). Based on the data, we arbitrarily defined a normal amount of cusp tissue as 18 mm or more (29); a lesser amount of tissue was considered retracted (29). Computer simulation studies (31) showed that the ideal cusp configuration existed when eH measured 0.45× gH, which was later confirmed by an anatomical study.
Based on this evolution, our routine assessment has been constant over the past 17 years. We used both gH and eH as objective measurements to determine the conduct of the operation; not only relying on visual assessment may be an explanation for the frequent detection (and correction) of cusp prolapse compared to other series (3,4,21,22,24). In fact, we saw a normal cusp configuration after completion of root remodeling in only 9% of the cases, while the remainder required some cusp repair (32).
A more recent modification has been the management of annular dilatation. Initially, we limited root remodeling to patients without relevant annular dilatation (5). This was based on the suggestion that a lack of annular stabilization after root remodeling was one of the leading mechanisms of late failure (3); also, others have attributed failures to the lack of annular size reduction or stabilization (33). Interestingly, however, the contribution of isolated annular dilatation in the absence of cusp prolapse has not been specified in any of the studies (21,22,24,33,34). In one series (33), the results were markedly improved by the addition of an annuloplasty and concomitant eH measurement.
Assuming that the annular diameter plays an important role in dilatation (31), we first introduced the concept of a suture annuloplasty to isolated aortic valve repair (35). An annuloplasty was also added to other forms of valve preserving procedures including root remodeling. Early results were promising in that the proportion of competent aortic valves at discharge increased significantly (14). However, the hardest evidence for the importance of annular stabilization on repair durability in our practice was obtained with isolated BAV repair (14,28,36). Previously, annular dilatation was an independent risk factor for failure in isolated BAV repair (28), and the stabilization and/or reduction of the annulus markedly and significantly improved repair durability (36,37). Later, we also found improved repair durability with UAVs and annuloplasty (10). We have, however, not yet seen an improvement in valve durability with the addition of annuloplasty when BAVs were treated by root remodeling (38,39). With TAV anatomy, the addition of an annuloplasty has not yet shown a significant effect on freedom from reoperation. This experience was surprising to us; it contradicted our expectations and the experience of others (34). A possible explanation for this observation could lie in the eH-driven and thus aggressive strategy of cusp repair. In doing so, we were able to treat a relevant number of root aneurysms with prolapse of all three cusps. The avoidance of symmetrical prolapse could perceivably reduce the stress at the level of the ring and thus contribute to annular size reduction (40). Of the different annuloplasty options (41), we used a suture annuloplasty for ease of application.
In assessing the current long-term experience, it has become increasingly clear that both adequate postoperative valve configuration and the original aortic valve anatomy (i.e., tricuspid, bicuspid or unicuspid) must be considered. This is confirmed by the current data. While visual assessment of adequate valve form is seemingly easier in BAV than in TAV, valves may still fail due to symmetric prolapse (26). With experience and longer follow-up, an increasing proportion of non-TAVs fails in the second decade due to calcification. The highest probability of failure is to be expected if pericardial patches have been used for cusp reconstruction (38,39). Based on the importance of commissural orientation for hemodynamics and durability of BAV repair (42), we modified the remodeling procedure to achieve a symmetric configuration whenever possible. This has resulted in even lower systolic gradients than we have observed previously; the creation of a TAV configuration is reserved for very asymmetric BAVs (Type C) (43). In UAVs, failure will occur even earlier and generally affects the pericardium used for cusp repair. In this context, it appears noteworthy that remodeling can be utilized to modify UAV anatomy in such a way that no patch material is necessary (17). Further follow-up will be required to judge the long-term value of this approach.
The current series includes different indications, i.e., aneurysm, severe AR, acute aortic dissection and connective tissue disease. Early mortality was low (1.5%) despite the inclusion of patients with acute aortic dissection (44). Similarly, we observed a low morbidity. Only 2.5% required surgical reintervention for hemorrhage, indicating that the procedure is as hemostatic as other procedures (4,45); this has been confirmed by a multi-center-analysis (45). Importantly, we did not observe postoperative atrioventricular block requiring pacemaker implantation, which differs from other results (46). The reason is most likely due to less basal dissection and the difference in suturing. Freedom from reoperation in the second decade was not affected by the presence of connective tissue disease (97% at 15 years with vs. 90% without). Similarly, no relevant difference was observed between patients with aneurysm as the primary indication vs. those with AR.
Secondary valve failure with early or late development of relevant AR was the most important valve-related complication observed (18,22,38). We previously identified cusp prolapse and low commissural height as the main causes of early failure after aortic valve reimplantation (47). In fact, persistent or recurrent prolapse was a predictor of failure in previous studies (40) and the majority of cases with postoperative cusp prolapse in our study had been performed prior to the introduction of intraoperative measurement of eH.
In addition, cusp pathology was the main reason for failure in most instances in a recent analysis of reoperations after remodeling (48); persistent/recurrent cusp prolapse or degeneration of patch material used for cusp repair were the main pathologies in some BAV and all UAV. In TAV, prolapse and secondary retraction were the most frequent mechanisms. Interestingly, low commissural height or isolated annular dilatation were not identified as reasons for failure (48). Valve calcification (2.5%; most had a BAV) and cusp retraction (0.65%) were relatively rare (48).
In the early experience, i.e., without eH measurement, cusp repair was performed in only a limited proportion of procedures (35%). In those, freedom from reoperation was identical in the first ten postoperative years irrespective of concomitant cusp repair. Only in the second decade, concomitant cusp repair was associated with inferior freedom from reoperation (P=0.207). With control of cusp configuration by eH measurement, identical results were obtained (P=0.407). This supports our research over the years that measurement of eH leads to better valve durability than visual assessment only.
Interestingly, we found inferior long-term survival in TAVs and absence of cusp plication; it did not have a significant effect on freedom from reoperation. Results were comparable when one, two or three cusps were repaired, both in survival and freedom from reoperation. Patients with prolapse repair of three cusps fared somewhat worse only regarding freedom from AR ≥2 at ten years.
While we have observed a few instances of annular dilatation in conjunction with cusp prolapse, no valve has failed yet solely due to annular dilatation. The presence of cusp prolapse may have a negative effect on annular stress distribution and its absence contributes to root stabilization. Thus, the current limited positive effects of an annuloplasty in our experience are not completely surprising. While we have found a clear stabilizing effect of a suture annuloplasty in isolated BAV repair (37), this positive effect was not observed with remodeling for BAV (39) and now for TAV. These findings correlate with a previous study in that even without annuloplasty, a size reduction of the annulus was observed (49). Nevertheless, an annuloplasty improves early valve competence; it may improve late durability of valve repair beyond the first 15 years and is thus probably a useful adjunct.
Conclusions
Root remodeling is a viable option in valve-preserving root replacement, both for tricuspid, bicuspid, and unicuspid valve morphologies. If combined with objective assessment of cusp configuration and aggressive cusp repair, reproducible and durable restoration of aortic valve function can be achieved.
Acknowledgments
Funding: None.
Footnote
Conflicts of Interest: The authors have no conflicts of interest to declare.
Open Access Statement: This is an Open Access article distributed in accordance with the Creative Commons Attribution-NonCommercial-NoDerivs 4.0 International License (CC BY-NC-ND 4.0), which permits the non-commercial replication and distribution of the article with the strict proviso that no changes or edits are made and the original work is properly cited (including links to both the formal publication through the relevant DOI and the license). See: https://creativecommons.org/licenses/by-nc-nd/4.0/.
References
- Sarsam MA, Yacoub M. Remodeling of the aortic valve anulus. J Thorac Cardiovasc Surg 1993;105:435-8. [Crossref] [PubMed]
- Yacoub MH, Gehle P, Chandrasekaran V, et al. Late results of a valve-preserving operation in patients with aneurysms of the ascending aorta and root. J Thorac Cardiovasc Surg 1998;115:1080-90. [Crossref] [PubMed]
- Erasmi AW, Sievers HH, Bechtel JF, et al. Remodeling or reimplantation for valve-sparing aortic root surgery? Ann Thorac Surg 2007;83:S752-6; discussion S785-90. [Crossref] [PubMed]
- David TE, Feindel CM, Webb GD, et al. Long-term results of aortic valve-sparing operations for aortic root aneurysm. J Thorac Cardiovasc Surg 2006;132:347-54. [Crossref] [PubMed]
- Schäfers H, Fries R, Langer F, et al. Valve-preserving replacement of the ascending aorta: remodeling versus reimplantation. J Thorac Cardiovasc Surg 1998;116:990-6. [Crossref] [PubMed]
- Langer F, Graeter T, Nikoloudakis N, et al. Valve-preserving aortic replacement: does the additional repair of leaflet prolapse adversely affect the results? J Thorac Cardiovasc Surg 2001;122:270-7. [Crossref] [PubMed]
- Langer F, Aicher D, Kissinger A, et al. Aortic valve repair using a differentiated surgical strategy. Circulation 2004;110:II67-73. [Crossref] [PubMed]
- Graeter TP, Fries R, Aicher D, et al. In-vitro comparison of aortic valve hemodynamics between aortic root remodeling and aortic valve reimplantation. J Heart Valve Dis 2006;15:329-35. [PubMed]
- Schäfers HJ, Kunihara T, Fries P, et al. Valve-preserving root replacement in bicuspid aortic valves. J Thorac Cardiovasc Surg 2010;140:S36-40; discussion S45-51. [Crossref] [PubMed]
- Franciulli M, Aicher D, Rädle-Hurst T, et al. Root remodeling and aortic valve repair for unicuspid aortic valve. Ann Thorac Surg 2014;98:823-9. [Crossref] [PubMed]
- Schäfers HJ, Bierbach B, Aicher D. A new approach to the assessment of aortic cusp geometry. J Thorac Cardiovasc Surg 2006;132:436-8. [Crossref] [PubMed]
- Bierbach BO, Aicher D, Issa OA, et al. Aortic root and cusp configuration determine aortic valve function. Eur J Cardiothorac Surg 2010;38:400-6. [Crossref] [PubMed]
- Graeter TP, Aicher D, Langer F, et al. Mid-term results of aortic valve preservation: remodelling vs. reimplantation. Thorac Cardiovasc Surg 2002;50:21-4. [Crossref] [PubMed]
- Aicher D, Schneider U, Schmied W, et al. Early results with annular support in reconstruction of the bicuspid aortic valve. J Thorac Cardiovasc Surg 2013;145:S30-4. [Crossref] [PubMed]
- Aicher D, Langer F, Kissinger A, et al. Valve-sparing aortic root replacement in bicuspid aortic valves: a reasonable option? J Thorac Cardiovasc Surg 2004;128:662-8. [Crossref] [PubMed]
- Schäfers HJ, Aicher D, Riodionycheva S, et al. Bicuspidization of the unicuspid aortic valve: a new reconstructive approach. Ann Thorac Surg 2008;85:2012-8. [Crossref] [PubMed]
- Matsushima S, Heß A, Glenske M, et al. Cusp Nadir Relocation by Root Remodeling in Unicuspid Aortic Valve Repair. Ann Thorac Surg 2019;108:e409-12. [Crossref] [PubMed]
- Leyh RG, Schmidtke C, Sievers HH, et al. Opening and closing characteristics of the aortic valve after different types of valve-preserving surgery. Circulation 1999;100:2153-60. [Crossref] [PubMed]
- Fries R, Graeter T, Aicher D, et al. In vitro comparison of aortic valve movement after valve-preserving aortic replacement. J Thorac Cardiovasc Surg 2006;132:32-7. [Crossref] [PubMed]
- Miyahara S, Karliova I, Giebels C, et al. Aortic root remodeling in bicuspid and tricuspid aortic valves-long-term results. Indian J Thorac Cardiovasc Surg 2020;36:81-7. [Crossref] [PubMed]
- Bethea BT, Fitton TP, Alejo DE, et al. Results of aortic valve-sparing operations: experience with remodeling and reimplantation procedures in 65 patients. Ann Thorac Surg 2004;78:767-72; discussion 767-72. [Crossref] [PubMed]
- Luciani GB, Casali G, Tomezzoli A, et al. Recurrence of aortic insufficiency after aortic root remodeling with valve preservation. Ann Thorac Surg 1999;67:1849-52; discussion 1853-6. [Crossref] [PubMed]
- Miyahara S, Omura A, Sakamoto T, et al. Impact of postoperative cusp configuration on midterm durability after aortic root reimplantation. J Heart Valve Dis 2013;22:509-16. [PubMed]
- Rankin JS, Gaca JG. Techniques of aortic valve repair. Innovations (Phila) 2011;6:348-54. [Crossref] [PubMed]
- Asano M, Kunihara T, Aicher D, et al. Mid-term results after sinutubular junction remodelling with aortic cusp repair. Eur J Cardiothorac Surg 2012;42:1010-5. [Crossref] [PubMed]
- Schäfers HJ, Aicher D, Langer F, et al. Preservation of the bicuspid aortic valve. Ann Thorac Surg 2007;83:S740-5; discussion S785-90. [Crossref] [PubMed]
- Tamer S, Mastrobuoni S, van Dyck M, et al. Free margin length and geometric height in aortic root dilatation and leaflet prolapse: implications for aortic valve repair surgery. Eur J Cardiothorac Surg 2020;57:124-32. [Crossref] [PubMed]
- Aicher D, Kunihara T, Abou Issa O, et al. Valve configuration determines long-term results after repair of the bicuspid aortic valve. Circulation 2011;123:178-85. [Crossref] [PubMed]
- Schäfers HJ, Schmied W, Marom G, et al. Cusp height in aortic valves. J Thorac Cardiovasc Surg 2013;146:269-74. [Crossref] [PubMed]
- De Kerchove L, Momeni M, Aphram G, et al. Free margin length and coaptation surface area in normal tricuspid aortic valve: an anatomical study. Eur J Cardiothorac Surg 2018;53:1040-8. [Crossref] [PubMed]
- Marom G, Haj-Ali R, Rosenfeld M, et al. Aortic root numeric model: annulus diameter prediction of effective height and coaptation in post-aortic valve repair. J Thorac Cardiovasc Surg 2013;145:406-411.e1. [Crossref] [PubMed]
- Ehrlich T, Hagendorff A, Abeln K, et al. Aortic cusp abnormalities in patients with trileaflet aortic valve and root aneurysm. Heart 2022;109:55-62. [Crossref] [PubMed]
- Lansac E, Di Centa I, Vojacek J, et al. Valve sparing root replacement: the remodeling technique with external ring annuloplasty. Ann Cardiothorac Surg 2013;2:117-23. [PubMed]
- de Kerchove L, Mastrobuoni S, Boodhwani M, et al. The role of annular dimension and annuloplasty in tricuspid aortic valve repair. Eur J Cardiothorac Surg 2016;49:428-37; discussion 437-8. [Crossref] [PubMed]
- Schäfers HJ, Aicher D. Root remodeling for aortic root dilatation. Ann Cardiothorac Surg 2013;2:113-6. [PubMed]
- Navarra E, El Khoury G, Glineur D, et al. Effect of annulus dimension and annuloplasty on bicuspid aortic valve repair. Eur J Cardiothorac Surg 2013;44:316-22; discussion 322-3. [Crossref] [PubMed]
- Schneider U, Hofmann C, Schöpe J, et al. Long-term Results of Differentiated Anatomic Reconstruction of Bicuspid Aortic Valves. JAMA Cardiol 2020;5:1366-73. [Crossref] [PubMed]
- Schneider U, Feldner SK, Hofmann C, et al. Two decades of experience with root remodeling and valve repair for bicuspid aortic valves. J Thorac Cardiovasc Surg 2017;153:S65-71. [Crossref] [PubMed]
- Froede L, Abeln KB, Ehrlich T, et al. Twenty-five years' experience with root remodeling and bicuspid aortic valve repair. Ann Cardiothorac Surg 2022;11:418-25. [Crossref] [PubMed]
- Kunihara T, Aicher D, Rodionycheva S, et al. Preoperative aortic root geometry and postoperative cusp configuration primarily determine long-term outcome after valve-preserving aortic root repair. J Thorac Cardiovasc Surg 2012;143:1389-95. [Crossref] [PubMed]
- Federspiel JM, Ehrlich T, Abeln K, et al. Aortic annuloplasty: Subcommissural, intra-annular suture techniques, external and internal rings. JTCVS Tech 2021;7:98-102. [Crossref] [PubMed]
- Froede L, Schäfers S, Wagenpfeil G, et al. Simplified determination of commissural orientation in bicuspid aortic valves. Eur J Cardiothorac Surg 2020;58:1153-60. [Crossref] [PubMed]
- de Kerchove L, Mastrobuoni S, Froede L, et al. Variability of repairable bicuspid aortic valve phenotypes: towards an anatomical and repair-oriented classification. Eur J Cardiothorac Surg 2019;ezz033. [Crossref] [PubMed]
- Ehrlich T, Karliova I, Froede L, et al. Aortic Root Remodeling in Acute Aortic Dissection. Thorac Cardiovasc Surg 2021;69:329-35. [Crossref] [PubMed]
- Chauvette V, Kluin J, de Kerchove L, et al. Outcomes of valve-sparing surgery in heritable aortic disorders: results from the AVIATOR registry. Eur J Cardiothorac Surg 2022;62:ezac366. [Crossref] [PubMed]
- Lansac E, Di Centa I, Sleilaty G, et al. Remodeling root repair with an external aortic ring annuloplasty. J Thorac Cardiovasc Surg 2017;153:1033-42. [Crossref] [PubMed]
- Giebels C, Aicher D, Kunihara T, et al. Causes and management of aortic valve regurgitation after aortic valve reimplantation. J Thorac Cardiovasc Surg 2013;145:774-80. [Crossref] [PubMed]
- Giebels C, Fister JC, Ehrlich T, et al. Failures of Valve-sparing Aortic Root Replacement Using the Root Remodeling Technique. Ann Thorac Surg 2022;113:2000-6. [Crossref] [PubMed]
- Kunihara T, Arimura S, Sata F, et al. Aortic annulus does not dilate over time after aortic root remodeling with or without annuloplasty. J Thorac Cardiovasc Surg 2018;155:885-894.e3. [Crossref] [PubMed]











