Origin of prefabricated frozen elephant trunk
Introduction
In the past two decades, thousands of patients presenting with acute or chronic aortic arch pathologies have been treated with an frozen elephant trunk (FET) graft. This technique includes implantation of a prefabricated Dacron graft, partially instrumented with a metal stent, which is inserted into the descending part of the thoracic aorta. Optimized versions of the medical product carry three branches to connect the supra-aortic vessels, a collar to facilitate its distal aortic anastomosis, plus an additional branch for temporary canulation to connect the “neo-aorta” to the extracorporeal circulation (ECC) system.
The origin of the prefabricated FET, as in clinical use today, was anything other than the spontaneous idea of an individual surgeon. It came as the result of numerous approaches by clinical researchers around the world, exploring the technology by intermediate solutions and self-fabricated implants. Their prime objective was to facilitate a one-step surgical approach to correcting aortic pathologies downstream from the ascending thoracic aorta and the aortic arch in order to avoid secondary procedures. The conventional two-stage treatment of extended aneurysms, atherosclerotic disorders or aortic dissections had resulted in significant interim mortality between the first and the second stage. In addition, a substantial operative mortality was noted with open aortic replacement.
The inaugurators of the first prefabricated FET had one prime objective, to improve the results of surgery for acute type A aortic dissection, both short and long term, and with a special focus on younger patients. Today, larger cohort (1-5) data from registries (6) and/or meta-analyses (7) published by various groups in different countries would suggest that the operative mortality has been reduced to about 10%, from about 30% three decades ago. National differences in these results may be related to patient selection and differences in healthcare systems.
The global success of the FET is, however, not the merit of surgical innovation alone. Without contemporary improvements in neighbouring technologies, which enabled and facilitated its use, the FET would never have become routine in clinical practice today.
Enabling and facilitating medical technology
Only five decades ago, there were no blood-tight vascular grafts available for thoracic aortic surgery, where full heparinization and ECC was needed. Fibrin sealant or albumin was often used to seal Dacron grafts immediately prior to their insertion, but many patients died from uncontrollable bleeding through the interstices of the knitted or woven fabric prostheses at that time. In the mid 1980s, blood-tight Dacron grafts pre-sealed by collagen or gelatine were offered commercially, and thoracic aortic surgery became much safer with respect to bleeding complications worldwide.
In the early days of surgery for aortic dissection, neither echocardiography nor computed tomography (CT) scans had been developed for clinical use. Thus, both aortography, using peripheral arterial cannulation, and coronary angiography were the standard preoperative imaging procedures to assure the correct diagnosis. Many patients died from rupture or cardiac tamponade during these invasive and time-consuming interventions, if presenting in the acute stage of the disease. CT-angiography, introduced into the routine to diagnose the site and extent of the dissecting process, facilitated the pre-operative work-up tremendously. This imaging modality quickly became available in many hospitals and became part of guidelines for preoperative diagnosis (8). Consequently, referrals for surgery for aortic dissections increased significantly in many countries over the past 30 years.
In the last years of the 20th century, the first stented grafts were explored for interventional percutaneous sealing of the entry site in acute type B dissections, or for internal coverage of atherosclerotic aortic lesions of limited extension within the descending thoracic aorta. Initially, their application as an intraoperative extension of the classic elephant trunk had not been envisioned, but a number of Japanese groups explored this innovative option (9,10). The fact, however, that not only radiologists, but also an increasing number of aortic surgeons became interested in this new technology, made the later step towards a prefabricated, one-piece device a logical and rather evolutionary step (Figure 1).
Worldwide, many patients presenting with aortic arch disease had to undergo a 50 years old and nearly abandoned concept of brain protection during cardiac surgery—deep hypothermic circulatory arrest. Using ECC, the body was cooled to approximately 20 degrees Celsius, preventing hypoxic brain damage for around 40 minutes. Concepts for cooling, warming, heparinization and bleeding control were discussed as major challenges for the conduct of ECC during such procedures. Yet, the risk of significant complications remained high, and innovative concepts for the conduct of ECC for aortic arch repair were intensely studied. The most devastating problem, hypoxic brain damage, underwent especially scrutinized experimental and clinical investigations.
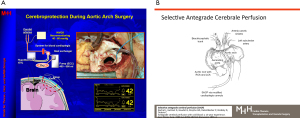
Protecting the brain from irreversible damage became a prime topic within the still limited group of aortic surgeons at the beginning of this century. In fact, this complication remained the main obstacle in any aggressive approaches in cases with distal extension of aortic arch disease. Today, we would consider antegrade cerebral perfusion (11-16), commonly via isolated cannulation of both carotid arteries, or retrograde perfusion via the superior caval vein (17), irreplaceable technologies for current approaches in surgical aortic arch replacement, including FET for complete repair (Figure 2).
Finally, an important basis for the later success of the FET were the numerous iterations of the initial ascending aortic repair to enable full arch repair in acute aortic dissections (Figure 3). Here, the driving force to achieve more complete repair was the high number of secondary operations after isolated repair of the ascending aorta, especially in younger patients with connective tissue disorders as underlying pathology for their aortic disorder (18). With this overarching objective in mind, the most decisive step was taken by Hans Borst in Hannover, Germany, by developing the original elephant trunk technique.
The elephant trunk procedure
In 1983, Borst (Figure 4) and his team published a paper describing their technique of the original elephant trunk application (19). Primarily designed to allow for easier extension of the arch prosthesis into the distal descending thoracic aorta during a second operation, it was later found to also be able to entirely correct limited proximal pathologies there. This concept was rapidly adopted by thoracic surgeon’s worldwide, and—like so many surgical innovations—was never submitted to a controlled clinical trial (20,21).
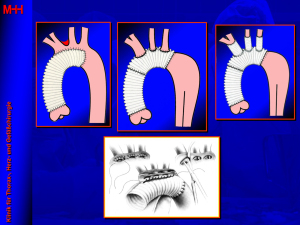
In brief, the technique involved a routine arch replacement using a straight Dacron graft, re-inserting the supra-aortic vessels, commonly via the island technique. The distal aortic anastomosis was performed first, but was performed in an end-to-side fashion rather than end-to-end, with the distal 6 cm being pushed into the proximal descending aorta. To facilitate and secure the side-to-side suture line distal to the subclavian artery opening, the proximal (arch) stretch of the graft was folded into the descending part of the prosthesis (Figure 5, left). During re-operation for later repair of the remaining pathology distally, the “trunk” portion of the graft could be easily grasped, clamped and extended by an additional Dacron tube (Figure 5, middle).
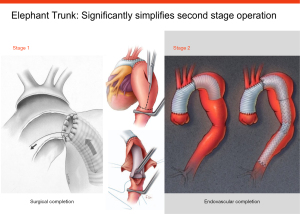
In response to the later development of stented Dacron grafts to correct isolated pathologies in the descending thoracic aorta, this innovative approach was then also utilized to extend the elephant trunk by interventional means, omitting a second open surgical repair (Figure 5, right).
The FET procedure
Two different routes on two continents were explored to assure distal exclusion of the aortic pathology by stent-grafting through the open aortic arch during the initial surgical procedure.
In Japan, a number of series were published, where the routine arch intervention, including concepts of antegrade or retrograde cerebral perfusion, was combined with the implantation of commercially available stent grafts into the descending thoracic aorta (9,10,22,23). The procedure was performed through median sternotomy and combined the concepts of the elephant trunk operation and endovascular stenting of descending aortic aneurysms (Figure 6).
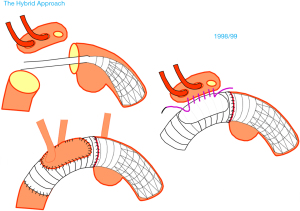
This hybrid approach resulted in a number of mobilised stent grafts during follow-up, even if suture fixation was attempted in the proximal descending thoracic aorta (23). Moreover, cases of paraplegia were seen, resulting in modifications of perfusion management during circulatory arrest (22). Nevertheless, several Japanese groups published a considerable number of case series, including a majority of patients with a successful outcome. To characterize this modification of the original elephant trunk technique, using two separate grafts, Ueda from Nagoya coined the term “Frozen elephant” procedure in 2002 (22).
Interestingly, his manuscript was probably in the editorial office already, when the Hannover group performed their first five cases using a self-made and prefabricated hybrid device, implanted as a single substitute (Figure 7). They had instrumented a stent into the distal segment of a Dacron tube made for aortic arch replacement. The inventor of the original elephant technique in Hannover, Hans Borst was asked what they should name the new development. Within 24 hours, he responded and suggested “Frozen Elephant Technique”. In retrospect, this appears to be a concomitant, geographically separated development for the same purpose, and named by the same term independently.

The first prefabricated elephant trunks were used in Germany in 2001 (24). Between September and November 2001, a total of five patients were operated on by the Hannover group (25). An early series of 10 patients was presented at an international meeting in 2002. The subsequent publication of 40 patients covered data on 40 procedures operated on until June 2002, and demonstrated obliteration of the peri-graft space (Figure 8). Subsequent studies described extended indications for the use of this custom-made graft, including an increasing number of patients operated on for acute aortic dissections (26,27).
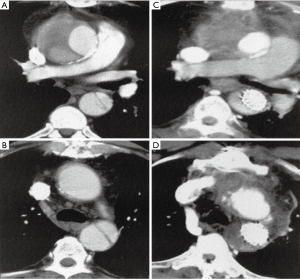
An important conceptual problem in this initial series was the fact that pre-sealing of stented grafts using collagen or gelatine was not possible at that time. Thus, the graft, which was distally stented, could not be used for arch replacement safely. The Hannover group used the “old” concept of pre-sealing the entire graft with Fibrin sealant (28).
A second obstacle occurred in patients with pathologies (usually dilatation) at the aortic segment downstream of the origin of the left subclavian artery, where the distal suture line of the arch portion of the prosthesis had to be constructed. Thus, a flexible collar made of soft Dacron for easy suturing was fabricated to be sewn to the arch portion of the graft at the level of the distal suture line without obstructing the orifice of the subclavian artery.
In addition, direct suturing of the branching vessels from the aortic arch into the Dacron tube when implanted as an arch replacement has always been difficult in diseased arteries, namely in aortic dissections involving these branches. Similarly, the island technique, as depicted in Figure 5, often resulted in imperfect sealing by sutures under such circumstances. Thus, a prefabricated graft for the aortic arch optimally includes three branches to connect these vessels. They can also be tailored in length and to fit the specific anatomic prerequisites of the individual patient, including the extent of his/her disease into their supra-aortic arteries.
Finally, difficulties were faced when inserting and deploying the stented portion of the arch graft into the open descending thoracic aorta, with a significant risk of injury to the aortic wall (Figure 9). Better engineering for this purpose was urgently required to provide a suitable sheath for insertion and a mechanism for deployment. A cuff made of soft Dacron positioned at the level of the distal aortic anastomosis was also included into the final design, as initially suggested in 2001 (Figure 10).
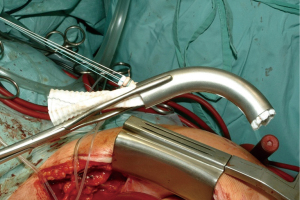
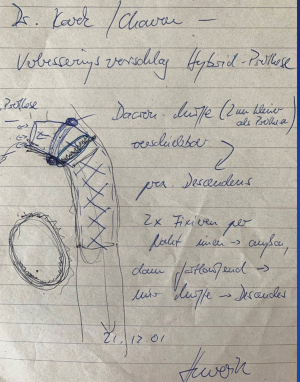
For the inaugurators, all these details were important for more widespread use of a facilitated procedure. After years of discussing with industry, a FET fulfilling these surgical requirements could be provided for pivotal clinical use. Following minor modifications, a design freeze was obtained, and a controlled clinical trial initiated. After successful completion, the FET, now a licensed medical product, was released for use in Europe in 2009. Many reports from various surgical groups confirmed its merits in complicated pathologies of the thoracic aorta involving the arch. However, complications were reported (29-32), resulting in refinement of patient selection and minor changes in the design of the product such as length of the stented portion of the graft. Nonetheless, the original idea to save patients requiring aortic arch repair from secondary surgical or interventional procedures had become reality, even in subjects presenting with acute aortic dissections.
Today, four different FET products are on the market. The first commercially available version of an FET was a non-branched version, with reports on early clinical results in 2005. This graft brings up an interesting anecdote Around the year 2000, the first author of this manuscript visited the founder and CEO of a medical product company, named Jostra, which no longer exists. The self-fabricated FET was presented with the aim to make it available outside his institution. The CEO gave a negative response. Later, the company was sold and renamed Jotec, and the still home-made device was presented to the new CEO, with a second negative response. The company was sold again, and inadvertently, a product came to market, suspiciously resembling the prototype from Hannover (Figure 11). This new company modified the graft repeatedly, including features like branches for the head vessels, and is now offered by a fourth company under a new name.
Comment
The prefabricated FET did not enter clinical practise by serendipity or disruptive innovation. It came as a result of a decade of international surgical attempts to improve both operative and long-term results in aortic arch surgery. The goal was cure of the patient by complete repair of the aorta in its diseased parts, where contemporary procedures could only offer palliation. Especially in younger patients presenting with aortic dissections, repair often left diseased segments of the aorta uncorrected, resulting in high rates of late death or re-operation (18).
The origin of the prefabricated FET came at a time that was ready for the intervention. Enabling technologies such as imaging, ECC, brain protecting measures and development of modern, stented vascular grafts had matured to a level where they could be utilized to lift aortic arch surgery to the next stage of surgical care. The fact that two distinct approaches on different continents both resulted in the development of a FET—with the same name independently—reflects the “technology readiness level” of aortic arch surgery.
Ultimately, any surgical innovation requires courage the part of both the surgeon and the patient. In this case, however, a third party has to be complimented on their courage—industry. It took the inaugurators many attempts, until finally, one company would enter the journey in prefabricating the FET. Twenty years later, four different products from different companies are on the market (33). For eligible patients, the prefabricated FET has evolved towards the gold standard in many aortic arch pathologies. Mortality has improved steadily with time. Accordingly, FET has found its way into national and international guidelines (34). It only took 25 years …
Acknowledgments
None.
Footnote
Funding: None.
Conflicts of Interest: The authors have no conflicts of interest to declare.
Open Access Statement: This is an Open Access article distributed in accordance with the Creative Commons Attribution-NonCommercial-NoDerivs 4.0 International License (CC BY-NC-ND 4.0), which permits the non-commercial replication and distribution of the article with the strict proviso that no changes or edits are made and the original work is properly cited (including links to both the formal publication through the relevant DOI and the license). See: https://creativecommons.org/licenses/by-nc-nd/4.0/.
References
- Shrestha M, Kaufeld T, Beckmann E, et al. Total aortic arch replacement with a novel 4-branched frozen elephant trunk prosthesis: Single-center results of the first 100 patients. J Thorac Cardiovasc Surg 2016;152:148-159.e1. [Crossref] [PubMed]
- Bojko MM, Oslund W, Kirsch MJ, et al. Commercial hybrid graft versus traditional arch replacement with frozen elephant trunk: A multi-institutional comparison. JTCVS Open 2025;23:19-33. [Crossref] [PubMed]
- Beckmann E, Martens A, Korte W, et al. Open total arch replacement with trifurcated graft and frozen elephant trunk. Ann Cardiothorac Surg 2020;9:170-7. [Crossref] [PubMed]
- Leone A, Beckmann E, Martens A, et al. Total aortic arch replacement with frozen elephant trunk technique: Results from two European institutes. J Thorac Cardiovasc Surg 2020;159:1201-11. Erratum in: J Thorac Cardiovasc Surg 2020;159:2571. [Crossref] [PubMed]
- Zhang K, Qiu J, Wu J, et al. Long-term outcomes in total arch replacement combined with frozen elephant trunk for acute type A aortic dissection. J Thorac Cardiovasc Surg 2024;S0022-5223(24)01106-1. [Epub ahead of print]. doi:
10.1016/j.jtcvs.2024.11.025 . - Okita Y. Current Status of Treatment for the Acute Type A Aortic Dissection in Japan. Semin Thorac Cardiovasc Surg 2025;37:155-64. [Crossref] [PubMed]
- Takagi H, Umemoto T. ALICE Group. A Meta-Analysis of Total Arch Replacement With Frozen Elephant Trunk in Acute Type A Aortic Dissection. Vasc Endovascular Surg 2016;50:33-46. [Crossref] [PubMed]
- Erbel R, Aboyans V, Boileau CESC Committee for Practice Guidelines, et al. 2014 ESC Guidelines on the diagnosis and treatment of aortic diseases: Document covering acute and chronic aortic diseases of the thoracic and abdominal aorta of the adult. The Task Force for the Diagnosis and Treatment of Aortic Diseases of the European Society of Cardiology (ESC). Eur Heart J 2014;35:2873-926. Erratum in: Eur Heart J 2015;36:2779. [Crossref] [PubMed]
- Kato M, Ohnishi K, Kaneko M, et al. New graft-implanting method for thoracic aortic aneurysm or dissection with a stented graft. Circulation 1996;94:II188-93.
- Usui A, Ueda Y, Watanabe T, et al. Clinical results of implantation of an endovascular covered stent-graft via midsternotomy for distal aortic arch aneurysm. Cardiovasc Surg 2000;8:545-9. [Crossref] [PubMed]
- Kazui T, Yamashita K, Washiyama N, et al. Usefulness of antegrade selective cerebral perfusion during aortic arch operations. Ann Thorac Surg 2002;74:S1806-9; discussion S1825-32. [Crossref] [PubMed]
- Bachet J, Guilmet D, Goudot B, et al. Antegrade cerebral perfusion with cold blood: a 13-year experience. Ann Thorac Surg 1999;67:1874-8; discussion 1891-4. [Crossref] [PubMed]
- Tian DH, Wan B, Bannon PG, et al. A meta-analysis of deep hypothermic circulatory arrest versus moderate hypothermic circulatory arrest with selective antegrade cerebral perfusion. Ann Cardiothorac Surg 2013;2:148-58. [Crossref] [PubMed]
- Okita Y, Miyata H, Motomura NJapan Cardiovascular Surgery Database Organization, et al. A study of brain protection during total arch replacement comparing antegrade cerebral perfusion versus hypothermic circulatory arrest, with or without retrograde cerebral perfusion: analysis based on the Japan Adult Cardiovascular Surgery Database. J Thorac Cardiovasc Surg 2015;149:S65-S73. [Crossref] [PubMed]
- Keeling WB, Leshnower BG, Hunting JC, et al. Hypothermia and Selective Antegrade Cerebral Perfusion Is Safe for Arch Repair in Type A Dissection. Ann Thorac Surg 2017;104:767-72. [Crossref] [PubMed]
- Hohri Y, Rajesh K, Murana G, et al. Safety of prolonged bilateral antegrade cerebral perfusion in aortic arch surgery with moderate hypothermia. Eur J Cardiothorac Surg 2024;67:ezae459. [Crossref] [PubMed]
- King RW, Carroll AM, Schäfer M, et al. High Flow, High-Pressure Retrograde Cerebral Perfusion at 28°C is Safe and Effective for Hemiarch Replacement of the Ascending Aorta. Aorta (Stamford) 2024;12:138-43. [Crossref] [PubMed]
- Haverich A, Miller DC, Scott WC, et al. Acute and chronic aortic dissections--determinants of long-term outcome for operative survivors. Circulation 1985;72:II22-34.
- Borst HG, Walterbusch G, Schaps D. Extensive aortic replacement using "elephant trunk" prosthesis. Thorac Cardiovasc Surg 1983;31:37-40. [Crossref] [PubMed]
- Bartels C, Gerdes A, Babin-Ebell J, et al. Cardiopulmonary bypass: Evidence or experience based? J Thorac Cardiovasc Surg 2002;124:20-7. [Crossref] [PubMed]
- Weber S, Haverich A. Pioneering surgical innovations in Germany: Part 1: generation of medical evidence. Chirurg 2016;87:423-32. [Crossref] [PubMed]
- Usui A, Fujimoto K, Ishiguchi T, et al. Cerebrospinal dysfunction after endovascular stent-grafting via a median sternotomy: the frozen elephant trunk procedure. Ann Thorac Surg 2002;74:S1821-4; discussion S1825-32. [Crossref] [PubMed]
- Usui A, Ueda Y, Akita T, et al. Mid-term results of an endovascular stent-graft by means of median sternotomy for distal aortic arch aneurysm. Artif Organs 2002;26:1044-9. [Crossref] [PubMed]
- Karck M, Chavan A, Hagl C, et al. The frozen elephant trunk technique: a new treatment for thoracic aortic aneurysms. J Thorac Cardiovasc Surg 2003;125:1550-3. [Crossref] [PubMed]
- Baraki H, Hagl C, Khaladj N, et al. The frozen elephant trunk technique for treatment of thoracic aortic aneurysms. Ann Thorac Surg 2007;83:S819-23; discussion S824-31. [Crossref] [PubMed]
- Pichlmaier MA, Teebken OE, Khaladj N, et al. Distal aortic surgery following arch replacement with a frozen elephant trunk. Eur J Cardiothorac Surg 2008;34:600-4. [Crossref] [PubMed]
- Ius F, Fleissner F, Pichlmaier M, et al. Total aortic arch replacement with the frozen elephant trunk technique: 10-year follow-up single-centre experience. Eur J Cardiothorac Surg 2013;44:949-57. [Crossref] [PubMed]
- Borst HG, Haverich A, Walterbusch G, et al. Fibrin adhesive: an important hemostatic adjunct in cardiovascular operations. J Thorac Cardiovasc Surg 1982;84:548-53.
- Nair JTK, Sathanantham DK, Chooraiyil N, et al. Mechanical Injury of Thoracic Aorta by Elephant Trunk Graft Limb following Frozen Elephant Trunk Procedure. Aorta (Stamford) 2024;12:98-101. [Crossref] [PubMed]
- Zhou C, Hou B, Zhang K, et al. Protective Effect on Spinal Cord Injury of Prophylactic Cerebrospinal Fluid Drainage in Extensive Aortic Arch Repair for Type A Aortic Dissection: A Retrospective Cohort Study. J Am Heart Assoc 2025;14:e039427. [Crossref] [PubMed]
- Karangelis D, Stougiannou TM, Christodoulou KC, et al. Hybrid Aortic Arch Replacement with Frozen Elephant Trunk (FET) Technique: Surgical Considerations, Pearls, and Pitfalls. J Clin Med 2024;13:7075. [Crossref] [PubMed]
- Tsagakis K, Kempfert J, Zierer A, et al. E-vita OPEN NEO in the treatment of acute or chronic aortic pathologies: first interim results of the NEOS study. Eur J Cardiothorac Surg 2024;65:ezae206. [Crossref] [PubMed]
- Hussain M, Jubouri YF, Hammad A, et al. The frozen elephant trunk: an overview of hybrid prostheses. Expert Rev Med Devices 2025;22:193-208. [Crossref] [PubMed]
- Czerny M, Schmidli J, Adler S, et al. Editor's Choice - Current Options and Recommendations for the Treatment of Thoracic Aortic Pathologies Involving the Aortic Arch: An Expert Consensus Document of the European Association for Cardio-Thoracic Surgery (EACTS) & the European Society for Vascular Surgery (ESVS). Eur J Vasc Endovasc Surg 2019;57:165-98. [Crossref] [PubMed]








