Sun’s procedure of total arch replacement using a tetrafurcated graft with stented elephant trunk implantation: analysis of early outcome in 398 patients with acute type A aortic dissection
Introduction
Despite increased understanding of the pathophysiology, improved anesthetic, myocardial and cerebral protection techniques, and advances in postoperative care of acute Stanford type A aortic dissection (AAAD) in the past two decades, AAAD remains one of the most lethal diseases in cardiovascular surgery and may arguably be increasing in incidence (1). Evidence for improving outcomes is modest (2) and mortality and neurological complication rates after surgical repair have been estimated to range from 15-30% (3,4). Although the frozen elephant trunk technique has greatly improved the clinical outcomes of AAADs with involvement of the arch vessels and proximal descending aorta, controversy regarding the validity of using this more aggressive approach in such critically ill patients still remains. The main counter-argument is that the higher technical complexity, longer operative time, and extended hypothermic circulatory arrest time may increase the mortality and morbidity, compared with standard repair at the distal ascending aorta level.
The Sun’s procedure is a surgical approach proposed by Dr. Li-Zhong Sun that integrates total arch replacement using a tetrafurcated graft with implantation of a special stented graft in the descending aorta (5,6). As of March 2013, our team has performed over 1,000 cases of Sun’s procedure with favorable early and late outcomes (6-12), among which there are 398 consecutive patients with AAAD. In this study, we seek to analyze the early outcomes of the Sun’s procedure in order to identify possible preoperative and intraoperative risk factors influencing the early mortality following emergent repair of AAADs, and attempt to provide evidence of the validity of the frozen elephant trunk procedure.
Patients and methods
The Ethics Committees of Fu Wai Hospital and Cardio-vascular Institute and Beijing Anzhen Hospital, Capital Medical University approved this retrospective study and waived the need for individual patient consent for this study.
Patients
Between April 2003 and March 2012, our team performed the Sun’s procedure in 398 patients with AAAD in Fu Wai Hospital (from April 2003 to December 2008; n=157) and Beijing Anzhen Hospital (from January 2009 to March 2012; n=241). There were 328 males and 70 females, with a mean age of 46±11 years (range, 17-78 years). The Sun’s procedure was selected for a patient with AAAD with (I) the primary entry located in the arch and descending aorta; or (IV) complicated with Marfan syndrome. Preoperatively, all patients were evaluated by computed tomographic angiography and echocardiography to confirm their diagnosis and assess the extent of dissection, diameters of aortic segments, evidence of malperfusion and sites of arterial cannulation. Table 1 lists 16 demographic and preoperative characteristics of the patients.
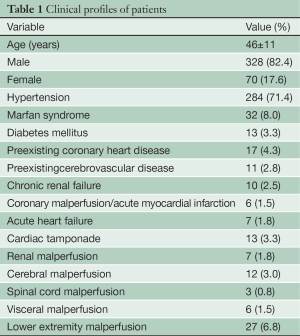
Full table
Surgical techniques
The Sun’s procedure refers to total arch replacement using a tetrafurcate vascular graft in combination with implantation of a special stented graft into the descending aorta. The technical details have been described in detail elsewhere (6-11) and in this issue of the Annals of Cardiothoracic Surgery (12). Briefly, it is performed with right axillary artery cannulation for cardiopulmonary bypass and selective antegrade cerebral perfusion, under moderate hypothermic circulatory arrest at 25 °C. This procedure involves implantation of the stented graft into the descending aorta, total arch replacement with a 4-branched vascular graft, a special sequence for aortic reconstruction (i.e., proximal descending aorta, then left carotid artery, ascending aorta, left subclavian artery, and finally innominate artery), and early rewarming and reperfusion after distal anastomosis to minimize cerebral and coronary ischemia. Selective cerebral perfusion time refers to the interval between the initiation of hypothermic circulatory arrest and completion of left carotid anastomosis, which is longer than the time of deep hypothermic circulatory arrest. In this period, lower body perfusion is arrested to implant the stented graft and suture the proximal descending anastomosis.
Statistical analysis
Patient demographic, preoperative, intraoperative, early morbidity and mortality data were collected and recorded prospectively. In this study, early mortality is defined as the percentage of patients undergoing the Sun’s procedure who die, including both (I) all deaths occurring during the same hospitalization after the primary surgery, even if after 30 days; and (II) deaths occurring after discharge from the hospital, but within 30 days of the procedure. Early morbidity is defined as any adverse events occurring within 30 days following the primary surgery.
Univariate analysis was used to examine the predictive values of 20 preoperative and intraoperative variables for early mortality: age, gender, hypertension, Marfan syndrome, diabetes mellitus, preexisting coronary artery disease, preexisting cerebrovascular disease, preexisting chronic renal failure, renal malperfusion, coronary malperfusion/acute myocardial infarction, acute heart failure secondary to aortic insufficiency or coronary ischemia, cardiac tamponade, cerebral malperfusion, spinal cord malperfusion, visceral malperfusion, lower limb malperfusion, location of primary intimal tear, and the times of cardiopulmonary bypass, aortic cross-clamp and selective cerebral perfusion. The correlation between these 20 variables and a composite end point of early mortality and morbidity including stroke, spinal cord injury, acute renal failure and additional surgical or endovascular interventions was also evaluated.
Statistical analysis was performed using SPSS for Windows software (version 13.0, Chicago, IL, USA). Continuous variables were expressed as mean ± standard deviation, and categorical variables were expressed as number (percentages). Comparisons between groups were analyzed with the chi-square test or Fisher’s exact test where appropriate. All variables associated with early mortality with a P value <0.1 in univariate analysis were examined consecutively with multivariate analysis using a forward stepwise binary logistic regression model. Any P value <0.05 was considered statistically significant.
Results
Operative data
The primary intimal tear was located at the ascending aorta in 178 cases (44.7%), transverse arch in 71 (17.8%), proximal descending aorta in 93 (23.4%), at multiple sites in 42 (10.6%) and was not identifiable in 14 (3.5%). Complete resection of intimal tear was achieved in all patients. The time of cardiopulmonary bypass, aortic cross-clamp and selective cerebral perfusion was 201±51 [103-442], 111±31 [52-279] and 24±8 [10-75] minutes respectively. Cannulation methods and concomitant procedures are presented in Table 2.
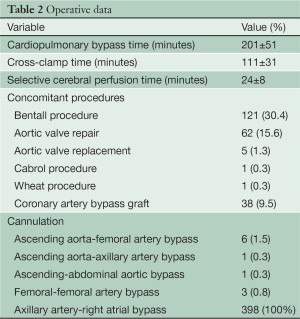
Full table
Early mortality and morbidity
Early mortality rate was 7.8% (31/398). The leading cause was multi-organ failure in 16 patients (51.6%, 16/31), permanent neurologic deficit (a new focal or global disorder of cerebral function lasting longer than 24 hours and still present at the time of hospital discharge or the patient’s death) in 6 (19.4%, 6/31), and low cardiac output syndrome in 4 (12.9%, 4/31). Other causes included renal failure (a rise in serum creatinine of 1 mg/dL above baseline or a new requirement of renal dialysis) in 2 patients (6.5%, 2/31), and aortic rupture, infection and coagulopathy, each in 1 (3.2%, 1/31).
Table 3 describes the 67 cases of early morbidities in detail (16.3%, 65/398), including renal failure in 17 (4.3%), permanent neurologic deficit and spinal cord injury, each in 10 (2.5%), low cardiac output and left recurrent nerve injury, each in 4 (1.0%), and limb ischemia in 2 (0.5%). Spinal cord injury included paraplegia in 7 patients (1.8%) and paraparesis in 3 (0.7%). Three patients complicated with permanent neurologic deficit were transferred for further neurologic treatment after their cardiopulmonary function stabilized.
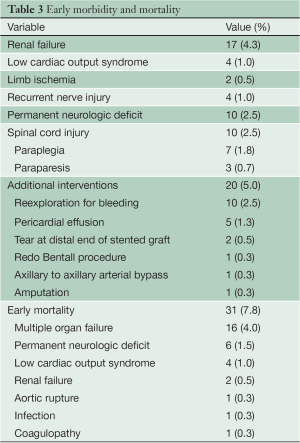
Full table
Additional surgical or endovascular procedures were required in 20 cases (5.0%), including reexploration for bleeding in 10 (2.5%), pericardial window for effusion in 5 (1.3%), endovascular stent grafting in 2 cases for large intimal tear at the distal end of the stented graft, redo Bentall procedure for persistent aortic root bleeding, axillary to axillary arterial bypass for left arm ischemia, and amputation for left leg thrombotic necrosis, each in 1 case.
Univariate analysis of potential risk factors for early mortality
Twenty preoperative and intraoperative variables were evaluated by univariate analysis for their predictive influence on early mortality (Table 4). Nine factors were remarkably associated with early mortality: preexisting cerebrovascular disease (P<0.001), renal malperfusion (P=0.012), coronary malperfusion/acute myocardial infarction (P=0.072), acute heart failure due to aortic insufficiency or coronary ischemia (P=0.012), cardiac tamponade (P=0.072), cerebral malperfusion (P=0.01), spinal cord malperfusion (P=0.017), visceral malperfusion (P<0.001) and cardiopulmonary bypass time >190 minutes (P=0.006).
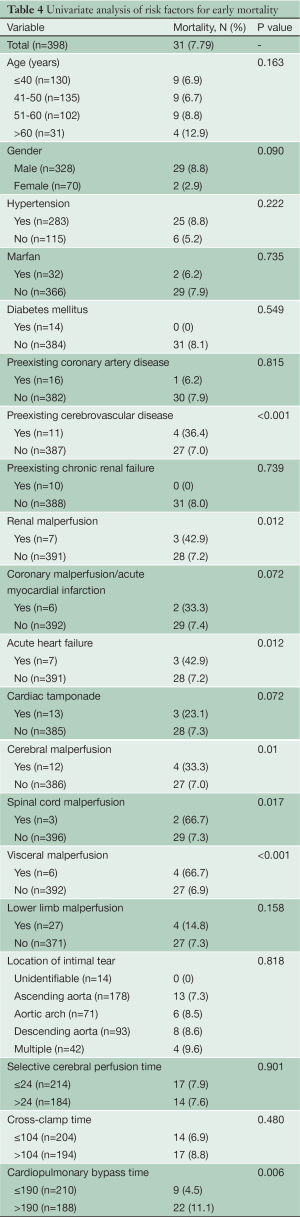
Full table
Multivariate analysis of potential risk factors for early mortality
Table 5 summarizes the results of multivariate analysis, by which five risk factors were identified to be independently associated with early postoperative mortality: preexisting cerebrovascular disease [relative risk (RR) =14.76; 95% confidence interval (CI), 3.61-60.06; P<0.001], acute heart failure secondary to aortic insufficiency or coronary ischemia (RR, 18.18; 95% CI, 3.44-96.04; P=0.001), spinal cord malperfusion (RR, 60.13; 95% CI, 4.60-786.95; P=0.002), visceral malperfusion (RR, 30.25; 95% CI, 4.93-185.60; P<0.001) and cardiopulmonary bypass time >190 minutes (RR, 3.62; 95% CI, 1.43-9.12; P=0.007).

Full table
Analysis of potential risk factors for early mortality and morbidity
When a composite end point including early mortality and morbidity was selected, univariate analysis found 8 significantly associated factors: preexisting cerebrovascular disease (P<0.001), renal malperfusion (P<0.001), cardiac tamponade (P=0.005), acute heart failure (P=0.072), cerebral malperfusion (P=0.024), spinal cord malperfusion (P<0.001), visceral malperfusion (P=0.001), lower limb malperfusion (P=0.023), and cardiopulmonary bypass time >190 minutes (P<0.001). In multivariate analysis, 4 risk factors were identified to be independently associated with early mortality and morbidity: preexisting cerebrovascular disease (RR, 4.06; 95% CI =1.10-14.98; P=0.035), renal malperfusion (RR, 29.74; 95% CI, 3.30-267.93; P=0.002), visceral malperfusion (RR, 7.25; 95% CI, 1.15-45.66; P=0.035) and cardiopulmonary bypass time >190 minutes (RR, 2.73; 95% CI, 1.52-4.92; P<0.001).
Discussion
Despite improvements in the surgical outcome of AAAD over the past two decades, postoperative mortality remains high. Although the frozen elephant trunk techniques have been shown to promote false lumen thrombosis, reduce distal patent false lumen rates, eliminate secondary tears in the arch and re-entry tears in the proximal descending aorta, ameliorate distal aortic malperfusion and decrease late aneurysm formation (8,13,14), there is still debate over its technical complexity, longer time of cerebral perfusion and hypothermic circulatory arrest, which might increase the risks of mortality and neurologic events (13,14). In addition, the indications of frozen elephant trunk beyond AAAD with descending aortic rupture (15) and retrograde dissection (9) are still controversial (16-18).
The objectives of this study are to analyze the early mortality and morbidity of the Sun’s procedure, (i.e., total arch replacement using 4-branched graft with implantation of a special stented graft in the descending aorta), in 398 consecutive patients with AAAD during a 10-year period, and to identify the risk factors predictive of early mortality and morbidity. Since large, prospective, randomized controlled clinical trials are ethically and practically infeasible, comparison of the results of Sun’s procedure with those of historical and contemporary series may be helpful in the choice of more aggressive frozen elephant trunk procedures versus the conservative standard repair for patients with AAAD.
Compared to operative mortality rates as high as 11-36% reported in the 1990s (19,20), the Sun’s procedure carries a relatively lower early mortality rate of 7.8% in 398 AAAD patients. Specifically, mortality rate in this series is also lower than that in most contemporary series of frozen elephant trunk procedure for AAAD patients, which ranged from 9.1-27.7% (21-24). This, in part, may be ascribed to the unique stented graft used in the Sun’s procedure (12) and an algorithm dictating the surgical strategies based on our sub-categorization of the Stanford system that divides type A dissections into subtypes according to the aortic root diameter, degree of aortic insufficiency, competency of the sinotubular junction, and the dissection process in the arch and beyond (10).
Another possible reason for the lower observed early mortality is the younger age of our patients (46±11 years). In contrast, data from the International Registry of Aortic Dissection (IRAD) suggest that one-third of patients with AAAD are over 70 years of age (25), and 640 of 2,137 patients (29.9%) in the German Registry for Acute Aortic Dissection Type A (GERAADA) are ≥70 years of age (26). Nevertheless, younger age seems to be a relative indication of the frozen elephant trunk procedure, as evidenced by the fact that the mean age of patients in recent cohorts ranged from 45.4-66.8 years, irrespective of geographic differences (21-24,27,28). To investigate the impact of age on early mortality of Sun’s procedure, we divided patients into 4 age groups and found a higher mortality rate in the group older than 60 years (12.9% versus 6.9%, 6.7%, 8.8% in other groups), but it did not reach statistical difference (Table 4). Multivariate analysis also failed to identify age as a predictor of early mortality in this study. Although this finding appears to be somewhat paradoxical and contrary to the common idea that old age is a risk factor, it is echoed in a report of 487 patients by Chiappini and associates (3). In a group of 355 AAAD patients, Kimura et al. (29) found that the early and late surgical outcomes were not better in 30 patients aged ≤45 years with a high risk of reoperation, which is similar to results of the IRAD study (30). Moreover, ascending aortic/hemiarch replacement was performed in at least 80% of Kimura’s patients (29), which implies, conversely, that the more aggressive frozen elephant trunk procedure should be chosen in younger patients.
Neurologic dysfunction after surgical repair of AAAD is a frequent complication resulting from brain or spinal cord ischemia. In the GERAADA study, the rate of neurologic dysfunctions was 20.3% (433/2,137) and 9.5% before and after surgery (31). The investigators found neurologic dysfunction was associated with increased risks of early mortality and identified extensive malperfusion, arch vessel dissection and longer operating time as risk factors of postoperative neurologic dysfunction. In this study, the incidence of neurologic dysfunction before and after surgery was 15.6% (62/398) and 5.0% (20/398) respectively, and permanent neurologic deficit caused 6 of 31 early deaths (19.4%). We also identified preoperative spinal cord ischemia as a risk factor of early mortality. In patients with intercostal arteries arising from the false lumen, spinal cord ischemia might be caused by rapid thrombosis of the false lumen, which implies that the spinal arterial collaterals cannot adequately compensate for acute spinal ischemia in patients with AAAD. Results of our study and GERAADA highlight the importance of early urgent surgical repair to restore brain and spinal cord blood supply and prevent such deficits from becoming permanent and deadly.
While the IRAD study demonstrated predictors of early mortality after surgical repair of AAAD to be pulse deficit, aortic rupture with preoperative hypotension, shock or cardiac tamponade, and signs of myocardial ischemia (32), we identified 5 risk factors for early mortality after the Sun’s procedure: preexisting cerebrovascular disease, acute heart failure secondary to aortic insufficiency or coronary malperfusion, spinal cord malperfusion, visceral malperfusion and cardiopulmonary bypass time >190 minutes. Renal malperfusion was also recognized as a risk factor with a composite end point of early mortality and morbidity. While prolonged cardiopulmonary bypass time is a well-defined risk factor for almost all open-heart procedures, 4 of these 6 risk factors belong to malperfusion syndrome, which has an incidence of 16-33% preoperatively (18). Malperfusion may result in myocardial, cerebral, spinal cord, extremity, renal and visceral ischemia and clinically apparent malperfusion of any type increases mortality risk (33). Pacini and associates reported a group of 502 patients and found that mesenteric malperfusion, coronary malperfusion and shock were risk factors for early mortality (34). In a group of 516 AAAD patients with coronary artery dissection, Imoto and colleagues demonstrated the mortality was 7.4% in those without ischemia versus 33% for those with ischemia (35). Our results corroborate other studies and suggest that immediate surgical repair to restore perfusion is vital to achieving a better outcome.
In conclusion, the Sun’s procedure has generated a relatively lower rate of early mortality and neurologic dysfunction in 398 patients with AAAD, compared to most contemporary and historical series. Patients with preexisting cerebrovascular disease, acute heart failure secondary to aortic sufficiency or coronary malperfusion, spinal cord malperfusion, visceral malperfusion and longer cardiopulmonary bypass time (>190 minutes) are at a higher risk of early mortality.
Acknowledgements
Disclosure: The authors declare no conflict of interest.
References
- Olsson C, Thelin S, Ståhle E, et al. Thoracic aortic aneurysm and dissection: increasing prevalence and improved outcomes reported in a nationwide population-based study of more than 14,000 cases from 1987 to 2002. Circulation 2006;114:2611-8. [PubMed]
- Olsson C, Eriksson N, Ståhle E, et al. Surgical and long-term mortality in 2634 consecutive patients operated on the proximal thoracic aorta. Eur J Cardiothorac Surg 2007;31:963-9; discussion 969. [PubMed]
- Chiappini B, Schepens M, Tan E, et al. Early and late outcomes of acute type A aortic dissection: analysis of risk factors in 487 consecutive patients. Eur Heart J 2005;26:180-6. [PubMed]
- Weigang E, Conzelmann LO, Kallenbach K, et al. German registry for acute aortic dissection type A (GERAADA)--lessons learned from the registry. Thorac Cardiovasc Surg 2010;58:154-8. [PubMed]
- Zhu XD. Preface. In: Sun LZ, Zhu JM, Liu YM, et al. eds. Surgery of the Aorta. 1st ed. Beijing: People’s Medical Publishing House, 2012:5.
- Ma WG, Zheng J, Liu YM, et al. Dr. Sun’s procedure for type A aortic dissection: total arch replacement using tetrafurcate graft with stented elephant trunk implantation. Aorta 2013;1:59-64.
- Liu ZG, Sun LZ, Chang Q, et al. Should the “elephant trunk” be skeletonized? Total arch replacement combined with stented elephant trunk implantation for Stanford type A aortic dissection. J Thorac Cardiovasc Surg 2006;131:107-13. [PubMed]
- Sun LZ, Qi RD, Chang Q, et al. Surgery for acute type A dissection using total arch replacement combined with stented elephant trunk implantation: experience with 107 patients. J Thorac Cardiovasc Surg 2009;138:1358-62. [PubMed]
- Sun L, Qi R, Chang Q, Zhu J, et al. Surgery for acute type A dissection with the tear in the descending aorta using a stented elephant trunk procedure. Ann Thorac Surg 2009;87:1177-80. [PubMed]
- Sun L, Qi R, Zhu J, et al. Repair of acute type a dissection: our experiences and results. Ann Thorac Surg 2011;91:1147-52. [PubMed]
- Sun L, Qi R, Zhu J, Liu Y, et al. Total arch replacement combined with stented elephant trunk implantation: a new “standard” therapy for type a dissection involving repair of the aortic arch? Circulation 2011;123:971-8. [PubMed]
- Ma WG, Zhu JM, Zheng J, et al. Sun’s procedure for complex aortic arch repair: total arch replacement using tetrafurcate graft with stented elephant trunk implantation. Ann Cardiothorac Surg 2013;2:642-8.
- Flores J, Kunihara T, Shiiya N, et al. Extensive deployment of the stented elephant trunk is associated with an increased risk of spinal cord injury. J Thorac Cardiovasc Surg 2006;131:336-42. [PubMed]
- Svensson LG. Device discordancy: lost cords, quick-fix seekers, quality, and ethics. J Thorac Cardiovasc Surg 2006;131:261-3. [PubMed]
- Sorokin VA, Chong CF, Lee CN, et al. Combined open and endovascular repair of acute type A aortic dissection. Ann Thorac Surg 2007;83:666-8. [PubMed]
- Bachet J. Icvts On-line discussion A. Patency of distal false lumen in acute dissection. Interact Cardiovasc Thorac Surg 2007;6:207-8. [PubMed]
- Murzi M, Glauber M. Should an endovascular procedure be combined with resection for type a aortic dissection? Ann Thorac Surg 2009;88:1387-8; author reply 1388-9. [PubMed]
- Bonser RS, Ranasinghe AM, Loubani M, et al. Evidence, lack of evidence, controversy, and debate in the provision and performance of the surgery of acute type A aortic dissection. J Am Coll Cardiol 2011;58:2455-74. [PubMed]
- Glower DD, Speier RH, White WD, et al. Management and long-term outcome of aortic dissection. Ann Surg 1991;214:31-41. [PubMed]
- Pansini S, Gagliardotto PV, Pompei E, et al. Early and late risk factors in surgical treatment of acute type A aortic dissection. Ann Thorac Surg 1998;66:779-84. [PubMed]
- Pochettino A, Brinkman WT, Moeller P, et al. Antegrade thoracic stent grafting during repair of acute DeBakey I dissection prevents development of thoracoabdominal aortic aneurysms. Ann Thorac Surg 2009;88:482-9; discussion 489-90. [PubMed]
- Jakob H, Tsagakis K, Pacini D, et al. The International E-vita Open Registry: data sets of 274 patients. J Cardiovasc Surg (Torino) 2011;52:717-23. [PubMed]
- Shen K, Tang H, Jing R, et al. Application of triple-branched stent graft for Stanford type A aortic dissection: potential risks. Eur J Cardiothorac Surg 2012;41:e12-7. [PubMed]
- Shrestha M, Pichlmaier M, Martens A, et al. Total aortic arch replacement with a novel four-branched frozen elephant trunk graft: first-in-man results. Eur J Cardiothorac Surg 2013;43:406-10. [PubMed]
- Trimarchi S, Eagle KA, Nienaber CA, et al. Role of age in acute type A aortic dissection outcome: report from the International Registry of Acute Aortic Dissection (IRAD). J Thorac Cardiovasc Surg 2010;140:784-9. [PubMed]
- Rylski B, Hoffmann I, Beyersdorf F, et al. Acute Aortic Dissection Type A: Age-related Management and Outcomes Reported in the German Registry for Acute Aortic Dissection Type A (GERAADA) of Over 2000 Patients. Ann Surg 2013. [Epub ahead of print]. [PubMed]
- Roselli EE, Rafael A, Soltesz EG, et al. Simplified frozen elephant trunk repair for acute DeBakey type I dissection. J Thorac Cardiovasc Surg 2013;145:S197-201. [PubMed]
- Uchida N, Katayama A, Tamura K, et al. Frozen elephant trunk technique and partial remodeling for acute type A aortic dissection. Eur J Cardiothorac Surg 2011;40:1066-71. [PubMed]
- Kimura N, Tanaka M, Kawahito K, et al. Early- and long-term outcomes after surgery for acute type a aortic dissection in patients aged 45 years and younger. Circ J 2011;75:2135-43. [PubMed]
- Januzzi JL, Isselbacher EM, Fattori R, et al. Characterizing the young patient with aortic dissection: results from the International Registry of Aortic Dissection (IRAD). J Am Coll Cardiol 2004;43:665-9. [PubMed]
- Conzelmann LO, Hoffmann I, Blettner M, et al. Analysis of risk factors for neurological dysfunction in patients with acute aortic dissection type A: data from the German Registry for Acute Aortic Dissection type A (GERAADA). Eur J Cardiothorac Surg 2012;42:557-65. [PubMed]
- Rampoldi V, Trimarchi S, Eagle KA, et al. Simple risk models to predict surgical mortality in acute type A aortic dissection: the International Registry of Acute Aortic Dissection score. Ann Thorac Surg 2007;83:55-61. [PubMed]
- Immer FF, Grobéty V, Lauten A, et al. Does malperfusion syndrome affect early and mid-term outcome in patients suffering from acute type A aortic dissection? Interact Cardiovasc Thorac Surg 2006;5:187-90. [PubMed]
- Pacini D, Leone A, Belotti LM, et al. Acute type A aortic dissection: significance of multiorgan malperfusion. Eur J Cardiothorac Surg 2013;43:820-6. [PubMed]
- Imoto K, Uchida K, Karube N, et al. Risk analysis and improvement of strategies in patients who have acute type A aortic dissection with coronary artery dissection. Eur J Cardiothorac Surg 2013;44:419-25. [PubMed]






