A meta-analysis of unmatched and matched patients comparing video-assisted thoracoscopic lobectomy and conventional open lobectomy
Background: Video-assisted thoracic surgery (VATS) for patients with early-stage non-small cell lung cancer (NSCLC) has been established as a safe and feasible alternative to open thoracotomy. This meta-analysis aims to assess the potential difference between unmatched and propensity score-matched cohorts who underwent VATS versus open thoracotomy in the current literature.
Methods: Three relevant studies with unmatched and propensity score-matched patients were identified from six electronic databases to examine perioperative outcomes after VATS lobectomy versus open thoracotomy for patients with early-stage NSCLC. Endpoints included perioperative mortality and morbidity, individual postoperative complications and duration of hospitalization.
Results: Results indicate that perioperative mortality was significantly lower for VATS compared to open thoracotomy in unmatched patients but no significant difference was detected amongst propensity score-matched patients. Similarly, the incidences of prolonged air leak and sepsis were significantly lower for VATS in the unmatched cohort, but not identified in the propensity score-matched cohort. In both the unmatched and matched groups, patients who underwent VATS were found to have a significantly lower overall perioperative morbidity rate, incidences of pneumonia and atrial arrhythmias, and a shorter duration of hospitalization in comparison to patients who underwent open thoracotomy.
Conclusions: The present meta-analysis indicates that VATS lobectomy has superior perioperative outcomes compared to open thoracotomy in both matched and unmatched cohorts. However, the extent of the superiority may have been overestimated in the unmatched patients when compared to propensity score-matched patients. Due to the limited number of studies with available data included in the present meta-analysis, these results are only of observational interest and should be interpreted with caution.
Key words: Video-assisted thoracic surgery; thoracotomy; non-small cell lung cancer; propensity score analysis; meta-analysis
Introduction
Similar to the introduction of laparoscopic appendicectomy in the 1980s, video-assisted thoracic surgery (VATS) for patients with non-small cell lung cancer (NSCLC) was pioneered in the early 1990s with great anticipation and enthusiasm. The benefits of this minimally invasive surgical technique has since shown encouraging perioperative outcomes in the form of reduced incidences of pneumonia (1), cardiac arrhythmias (2) and pain (3) compared to open thoracotomy. A recent meta-analysis suggests improved outcomes for VATS in terms of systemic recurrence and 5-year overall survival when compared to open thoracotomy for selected patients with early stage NSCLC (4).
Despite the multitude of reported superior short- and long-term outcomes in retrospective observational studies, the acceptance of VATS within the thoracic community has been slow. Currently, only a small fraction of pulmonary resections are performed by VATS globally. There is a paucity of robust clinical data in the form of large randomized controlled trials to compare VATS to open thoracotomy, and publication bias in the vast majority of retrospective studies in the existing literature cannot be excluded. Critics of VATS argue that the non-randomized patient selection process in retrospective studies may provide a ‘false positive’ finding of superior outcomes for VATS when more favorable patients are selected for this novel technique. Indeed, no randomized controlled trial has ever been completed to compare conventional VATS according to the current accepted Cancer and Leukemia Group B (CALGB) definition with open thoracotomy (5-7). To assess the potential patient selection bias for VATS in the current literature, we compared perioperative outcomes in unmatched patients with propensity score-matched patients to identify any significant differences between these two study cohorts.
Methods
Literature search strategy
Electronic searches were performed using PubMed, Ovid Medline, Cochrane Central Register of Controlled Trials, Cochrane Database of Systematic Reviews, ACP Journal Club, and Database of Abstracts of Review of Effectiveness from their date of inception to April 2012. We combined the terms “video-assisted thoracic surgery” or “VATS” or “thoracoscopic surgery” with “propensity*” or “propensity score” or “propensity match” as key words or MeSH terms. The reference lists of all retrieved articles were reviewed for further identification of potentially relevant studies. Eligible comparative studies for the present meta-analysis included those in which perioperative data were available for unmatched and propensity score-matched patients with NSCLC who underwent VATS or open thoracotomy.
All data were extracted from article texts, tables and figures. Two investigators (C.C. and S.A.) independently reviewed each retrieved article. Discrepancies between the two reviewers were resolved by discussion and consensus. The final results were reviewed by the senior investigators (C.M. and T.D.Y.).
Statistical analysis
The propensity score is the conditional probability of assignment to a particular treatment given a vector of observed covariates (8). This statistical method aims to minimize bias in retrospective observational studies by matching the individual’s measured covariates between two treatment groups, so that differences in the measured outcome can be more directly attributed to the treatment rather than the individual’s observed covariates (9). Propensity score matching is considered to significantly strengthen observational studies (8-10). Studies included in the present meta-analysis provided comparative data on perioperative outcomes for unmatched and propensity score-matched patients who underwent VATS or open thoracotomy. Meta-analysis was performed by combining the results of reported incidences of postoperative mortality, postoperative morbidity, individual postoperative complications and duration of hospitalization for unmatched patients. The same process was then performed for the propensity score-matched patients.
The relative risk (RR) was used as a summary statistic. In the present study, the random effect models were tested, where it was assumed that there were variations between studies and the calculated ratios thus had a more conservative value (11). X2 tests were used to study heterogeneity between trials. I
Results
Quantity and quality of trials
Nineteen potentially relevant references were identified through the six electronic database searches. After exclusion of duplicate or irrelevant references, 7 potentially relevant articles were retrieved for more detailed evaluation (13-19). After applying the selection criteria, three comparative studies remained for assessment (13-15). Manual search of the reference lists did not identify any additional relevant studies. All 3 studies included for final analysis in the present meta-analysis were from retrospective observational studies. In these 3 studies, 7,730 unmatched patients with NSCLC were compared, including 5,636 patients who underwent open thoracotomy and 2,094 patients who underwent VATS. After propensity score-matching, these same 3 studies reported perioperative outcomes on 1,681 patients who underwent open thoracotomy with 1,681 patients who underwent VATS.
Assessment of perioperative mortality and morbidity
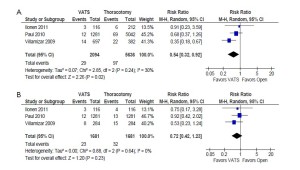
From the three selected studies, the overall perioperative mortality rate of unmatched patients was significantly lower in patients who underwent VATS compared to patients who underwent open thoracotomy (1.4% vs. 1.7%; RR, 0.54; 95% confidence interval [CI], 0.32-0.92; P=0.02; I2=30%). In comparison, propensity score-matched patients from the same studies reported a statistically non-significant difference in mortality rate between the two treatment groups (1.4% vs. 1.9%; RR, 0.72; 95% CI, 0.42-1.23; P=0.23; I2=0%). These results are summarized in Figure 1.
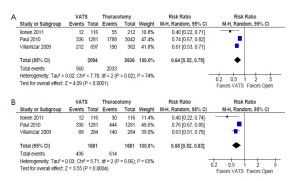
Overall perioperative morbidity rates were consistently reported to be significantly lower after VATS compared to open thoracotomy in both unmatched patients (26.7% vs. 36.1%; RR, 0.64; 95% CI, 0.52-0.79; P<0.0001; I2=74%) and propensity score-matched patients (25.9% vs. 36.5%; RR, 0.65; 95% CI, 0.52-0.83; P=0.0004; I2=65%). These results are summarized in Figure 2.
Assessment of postoperative complications
A number of postoperative complication rates were comparable between the three selected studies for both unmatched and propensity score-matched patients. These included prolonged air leak, pneumonia, pulmonary embolism, atrial arrhythmias, significant bleeding, empyema and sepsis. Patients who underwent VATS were found to have significantly lower incidences of pneumonia and atrial arrhythmias in both the unmatched and propensity score-matched cohorts.
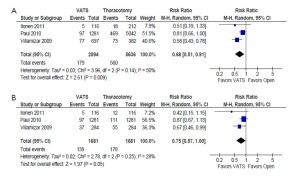
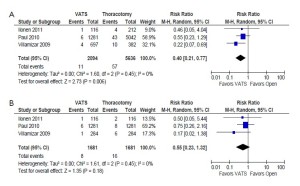
In the unmatched cohort, patients who underwent VATS were reported to have a statistically significantly lower incidence of prolonged air leak (8.5% vs. 9.9%; RR, 0.68; 95% CI, 0.51-0.91; P=0.009; I2=50%) and sepsis (0.5% vs. 1.0%; RR, 0.40; 95% CI, 0.21-0.77; P=0.006; I2=0%) compared to patients who underwent thoracotomy. However, these outcomes were not statistically significant when propensity score-matched patients were compared in the same studies. These results are summarized in Figure 3 and 4, respectively.
Assessment of length of hospitalization
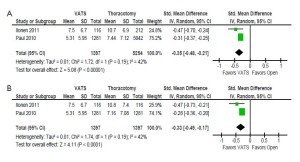
Comparable data for the duration of hospitalization was reported in two studies (13,15). VATS was found to be associated with a significantly shorter period of hospitalization for both unmatched patients (standardized mean difference –0.35; 95% CI –0.48- –0.21; P<0.00001; I2=42%) and propensity score-matched patients (standardized mean difference -0.33; 95% CI –0.49- –0.17; P<0.0001; I2=42%). These results are summarized in Figure 5 and an overall summary of perioperative outcomes for unmatched and propensity score-matched patients who underwent VATS versus open thoracotomy is presented in Table 1.
| Table 1 Summary of perioperative outcomes of unmatched and propensity score-matched patients who underwent video-assisted thoracic surgery (VATS) versus open thoracotomy for non-small cell lung cancer in three selected studies | ||
| Measured outcome | Unmatched patients | Matched patients |
| VATS n=2,094 Open n=5,636 | VATS n=1,681 Open n=1,681 | |
| Perioperative mortality | VATS | NS |
| Perioperative morbidity | VATS < Open | VATS < Open |
| Prolonged air leak | VATS < Open | NS |
| Pneumonia | VATS < Open | VATS < Open |
| Pulmonary embolism | NS | NS |
| Atrial arrhythmias | VATS < Open | VATS < Open |
| Bleeding | NS | NS |
| Empyema | NS | NS |
| Sepsis | VATS < Open | NS |
| Length of stay | VATS < Open | VATS < Open |
| ‘<’ indicates statistically lower rate or duration according to meta-analysis; NS, not significant | ||
Discussion
To date, the highest level of clinical evidence comparing VATS with open thoracotomy have been from retrospective observational studies. Two small randomized controlled trials compared patients with NSCLC who underwent VATS lobectomy versus open thoracotomy (6,7). However, it should be acknowledged that rib-spreading was performed in both reports and thus these studies no longer conform to the current definition of ‘true’ VATS lobectomies (5-7). The study conducted by Kirby et al. in 1995 randomized 61 patients with clinical stage I NSCLC to undergo VATS lobectomy or open thoracotomy (6). This study reported that VATS was associated with significantly fewer postoperative complications but not a significant decrease in blood loss, duration of chest tube drainage, length of hospital stay, or postoperative pain. A second study by Sugi and colleagues randomized 100 patients with clinical stage IA NSCLC to undergo VATS lobectomy or open lobectomy (7). This trial found no significant differences in recurrence or survival rates between the two treatment groups. It is important to note that patients randomized to the VATS arm in both studies were not analyzed according to an intention-to-treat approach, and patients intended for VATS but converted to open thoracotomy were included in the open thoracotomy group or excluded altogether from statistical analysis (20).
In recent years, a number of retrospective studies have utilized propensity score-matching as a statistical tool to minimize patient selection bias between VATS and open thoracotomy treatment groups (13-19). Flores and colleagues compared 313 propensity score-matched patients who underwent VATS lobectomy or open thoracotomy, and reported a similar 5-year overall survival, but significantly fewer postoperative complications and a shorter duration of hospitalization for VATS patients when compared to open thoracotomy. Unfortunately, detailed data were not available to be included in the present meta-analysis. Scott et al. reported two studies which used propensity score to compare patients undergoing VATS versus open thoracotomy (18,19). However, individuals were categorized into propensity score groups rather than being case-matched according to a 1:1 ratio, with ‘outlier’ patients who underwent thoracotomy being excluded from analysis (18). Park et al. recently published a study that provided data on 136 propensity score-matched patients and found VATS to be associated with a significantly shorter duration of hospitalization (16). However, no data was presented for unmatched patients in their cohort so this study was also excluded from the present meta-analysis.
Within the current literature, three studies were found to provide data on perioperative outcomes for both unmatched and propensity score-matched patients who underwent VATS versus open thoracotomy. Results from the present meta-analysis indicate that unmatched patients from these studies were likely to report more significant benefits after VATS compared to open thoracotomy. Specifically, unmatched patients who underwent VATS were found to have superior overall perioperative mortality and morbidity rates, as well as lower incidences of prolonged air leak, pneumonia, atrial arrhythmias and sepsis. In addition, the duration of hospitalization was significantly shorter after VATS compared to open surgery. In comparison, patients who were matched according to propensity score analysis in the same 3 studies did not show statistically significant difference in overall postoperative mortality and incidences of prolonged air leak and sepsis. These results may suggest that unmatched patients in the three included retrospective observational studies have overestimated the potential perioperative benefits of VATS compared to open thoracotomy. However, it should be acknowledged that the propensity score-matching process invariably reduces the number of patients included in the data analysis, which may decrease the statistical power of the comparative studies. In addition, due to the small number of the studies included in the present analysis, these results can only be regarded as an interesting observation, and should be interpreted with care.
There is growing evidence to suggest that VATS lobectomy is a safe and feasible operation associated with improved perioperative outcomes compared to conventional open thoracotomy (4,13,20). VATS lobectomy may be superior to open lobectomy in terms of oncologic efficacy based on advantages seen in surgical outcomes after thoracoscopic resections, and the delivery of adjuvant chemotherapy (21). As a result of these findings, the VATS lobectomy technique has become an addition to the armamentarium of many modern thoracic surgeons. However, it is necessary to appreciate that there are currently no randomized controlled trials comparing ‘true’ VATS lobectomy as defined by the CALGB criteria versus the open technique (5). Due to a lack of clinical equipoise and widely reported benefits of the minimally invasive approach, we may have missed the opportunity to conduct such randomized controlled trials. Despite the large number of retrospective comparative studies over the past two decades, the favored surgical approach for early-stage NSCLC remains controversial. From a different perspective, attention should perhaps be directed at the patient selection process to identify the most appropriate patients who will gain the maximal benefit from a minimally invasive approach. More emphasis should also be placed on the use of standardized, objective and reproducible outcome measures to provide a more reliable estimate of how much benefit can be offered to patients undergoing VATS. This may consolidate the role of VATS in lung cancer management, and may raise the standards of outcome measurement in thoracic surgery as a whole.
The present meta-analysis indicates that VATS lobectomy has superior perioperative outcomes compared to open thoracotomy in both matched and unmatched cohorts. However, the extent of these reported superior outcomes may be overestimated in the unmatched patients when compared with propensity score-matched patients.
Acknowledgements
Disclosure: The authors declare no conflict of interest.
References
- KWhitson BA, Andrade RS, Boettcher A, et al. Video-assisted thoracoscopic surgery is more favorable than thoracotomy for resection of clinical stage I non-small cell lung cancer. Ann Thorac Surg 2007;83:1965-1970.
- Muraoka M, Oka T, Akamine S, et al. Video-assisted thoracic surgery lobectomy reduces the morbidity after surgery for stage I non-small cell lung cancer. Jpn J Thorac Cardiovasc Surg 2006;54:49-55.
- Yim AP, Wan S, Lee TW, et al. VATS lobectomy reduces cytokine responses compared with conventional surgery. Ann Thorac Surg 2000;70:243-247.
- Yan TD, Black D, Bannon PG, et al. Systematic review and meta-analysis of randomized and nonrandomized trials on safety and efficacy of video-assisted thoracic surgery lobectomy for early-stage non-small-cell lung cancer. J Clin Oncol 2009;27:2553-2562.
- Swanson SJ, Herndon JE, D’Amico TA, et al. Video-assisted thoracic surgery lobectomy: report of CALGB 39802--a prospective, multi-institution feasibility study. J Clin Oncol 2007;25:4993-4997.
- Kirby TJ, Mack MJ, Landreneau RJ, et al. Lobectomy--video-assisted thoracic surgery versus muscle-sparing thoracotomy. A randomized trial. J Thorac Cardiovasc Surg 1995;109(5):997-1001.
- Sugi K, Kaneda Y, Esato K. Video-assisted thoracoscopic lobectomy achieves a satisfactory long-term prognosis in patients with clinical stage IA lung cancer. World J Surg 2000;24(1):27-30.
- Rosenbaum PR, Rubin DB. The central role of propensity score in observational studies for causal effects. Biometrika 1983;70:41-55.
- Blackstone EH. Comparing apples and oranges. J Thorac Cardiovasc Surg2002;123:8-15.
- D’Agostino RB, Jr. Propensity scores in cardiovascular research. Circulation 2007;115:2340-2343.
- DerSimonian R, Laird N. Meta-analysis in clinical trials. Controlled Clinical Trials 1986;7:177-188.
- Higgins JP, Thompson SG. Quantifying heterogeneity in a meta-analysis. Statistics in Medicine 2002;21:1539-1558.
- Paul S, Altorki NK, Sheng S, et al. Thoracoscopic lobectomy is associated with lower morbidity than open lobectomy: a propensity-matched analysis from the STS database. J Thorac Cardiovasc Surg 2010;139:366-378.
- Villamizar NR, Darrabie MD, Burfeind WR, et al. Thoracoscopic lobectomy is associated with lower morbidity compared with thoracotomy. J Thorac Cardiovasc Surg 2009;138:419-425.
- Ilonen IK, Rasanen JV, Knuuttila A, et al. Anatomic thoracoscopic lung resection for non-small cell lung cancer in stage I is associated with less morbidity and shorter hospitalization than thoracotomy. Acta oncologica (Stockholm, Sweden) 2011;50:1126-1132.
- Park JS, Kim K, Choi MS, et al. Video-Assisted Thoracic Surgery (VATS) Lobectomy for Pathologic Stage I Non-Small Cell Lung Cancer: A Comparative Study with Thoracotomy Lobectomy. The Korean journal of thoracic and cardiovascular surgery 2011;44:32-38.
- Flores RM, Park BJ, Dycoco J, et al. Lobectomy by video-assisted thoracic surgery (VATS) versus thoracotomy for lung cancer. J Thorac Cardiovasc Surg 2009;138:11-18.
- Scott WJ, Allen MS, Darling G, et al. Video-assisted thoracic surgery versus open lobectomy for lung cancer: a secondary analysis of data from the American College of Surgeons Oncology Group Z0030 randomized clinical trial. J Thorac Cardiovasc Surg 2010;139:976-981.
- Scott WJ, Matteotti RS, Egleston BL, et al. A comparison of perioperative outcomes of video-assisted thoracic surgical (VATS) lobectomy with open thoracotomy and lobectomy: results of an analysis using propensity score based weighting. Annals of surgical innovation and research 2010;4:1.
- Cao CQ, Stine ML, Yan TD. True video-assisted thoracic surgery for early-stage non-small cell lung cancer. J Thorac Dis 2009;1:34-8.
- Flores RM, Ihekweazu UN, Rizk N, et al. Patterns of recurrence and incidence of second primary tumors after lobectomy by means of video-assisted thoracoscopic surgery (VATS) versus thoracotomy for lung cancer. J Thorac Cardiovasc Surg 2011;141:59-64.






