Single-port thymectomy using a subxiphoid approach—surgical technique
Clinical vignette
Single-port thymectomy (SPT) with a subxiphoid approach is indicated for anterior mediastinal tumors and myasthenia gravis when preoperative diagnostic imaging reveals no tumor infiltration into the surrounding tissues, such as the brachiocephalic vein and pericardium. The present case is a 42-year-old female patient who had an incidental finding of an anterior mediastinal cystic mass during follow-up of asthma. The mass was 1 cm in diameter and did not infiltrate into nearby organs (Video 1).

Surgical technique
Preparation
With the patient in the supine position, SPT was performed with the surgeon standing between the patient’s legs and an assistant standing on the patient’s right ride to operate the camera. Differential lung ventilation was not necessary. A 30-degree, 5-mm ridged camera scope was used. When using a 5-mm camera scope, the safety of the surgery should be ensured by selecting an instrument that has sufficient lighting to prevent the operative field from darkening. The use of a long-length scope can prevent interference between the camera head and the surgeon’s hands (Figure 1).
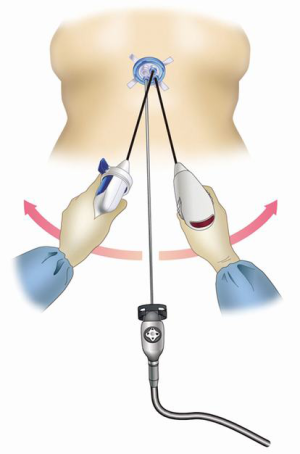
Exposition
First, a 3-cm transverse incision was made 1 cm caudal to the xiphoid process. After blind detachment of the sternum using a finger, a 0.5-cm longitudinal incision was made on the fascia of the rectus abdominis to create enough space to insert a GelPOINT mini advanced access platform (Applied Medical, Rancho Santa Margarita, CA, USA) for single incision surgery. If the skin incision is made too close to the xiphoid process, it becomes difficult for surgical instruments, such as vessel sealing devices, to reach the posterior aspect of the sternum. In such a scenario, the thymus may be separated from the sternum using roticulator scissors with a flexible tip.
Operation
One of three mini-ports inserted into the main port was used for the camera scope. CO2 insufflation in the mediastinum was performed at 8 mmHg. The positive pressure of CO2 insufflation, together with detachment of the thymus from the sternum, created a space sufficient for the surgeon to wield the Autonomy grasper (Cambridge Endo, Framingham, MA, USA) in the left hand and the LigaSure™ Maryland vessel sealing system (Covidien, Mansfield, MA, USA) in the right hand. The mediastinal pleura were opened bilaterally. Surgeons may experience difficulty in moving the tip of the vessel sealing system to its correct position due to interference from the camera scope or forceps. In single-port surgery, the camera scope and other surgical instruments cross over each other. With the single port system, the position of the tip of the vessel sealing system in the thoracic cavity varies greatly depending on whether the vessel sealing system is inserted above or below the camera scope or forceps through the single port. When the tip of the vessel sealing system does not move in the intended direction, it is necessary to change the way the vessel sealing system is inserted, depending on its position above or below the other instrument. To detach the right lobe of the thymus, the thymus is pulled towards the left side of the patient by directing the forceps to the left. To detach the left lobe, the thymus is pulled towards the right side of the patient by directing the forceps to the right. At this time, it is necessary for the surgeon to cross hands (Figure 2A,B). For detachment in the neck region, grasper forceps are used to grab and pull the superior pole of the thymus towards the caudal end in order to push the brachiocephalic vein and create a good operative field. The thymic vein is then dealt with. Because the path of the thymic vein is tangential to the vessel sealing system, the course of the thymic vein will run in a transverse direction on lateral traction of the thymus, which allows the vessel sealing system to separate the thymic vein from the brachiocephalic vein.
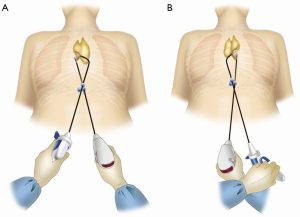
Completion
The resected thymus was placed in a widely opened pouch in the mediastinum and allowed to gravitate towards the bottom of the pouch, which was then retrieved through the subxiphoid incision. A 20-Fr drainage catheter was inserted through the same opening into the anterior mediastinum before completion of the surgery.
Comments
Clinical results
The length of operation was 123 minutes and blood loss was minimal. No complications occurred during or after surgery, and the patient was discharged from hospital four days after surgery. Thymic cyst was diagnosed based on pathological findings.
Advantages
In recent years, minimally invasive approaches have replaced conventional median sternotomy in thymectomy for thymoma or myasthenia gravis (1). The lateral thoracoscopic approach that is most commonly used in many institutions has several drawbacks, including difficulty in identifying the contralateral phrenic nerve and ensuring a sufficient operative field in the neck region. The lateral thoracoscopic approach is also associated with postoperative pain and numbness due to intercostal nerve impairment caused by incisions made in the intercostal space. Because the camera is inserted along the midline of the body, SPT (2), in contrast, provides a sufficient operative field in the neck region and makes the identification of both phrenic nerves easy. In addition, the SPT does not involve the intercostal space, thereby avoiding intercostal nerve damage. Furthermore, SPT is aesthetically excellent and minimally invasive as only a single 3 cm incision is made on the abdomen.
Caveats
One of the drawbacks of SPT is poor surgical manipulability because surgical instruments inserted in a single port interfere with each other. Therefore, we do not recommend SPT for cases requiring sophisticated surgical manipulation, such as those with tumor infiltration into the phrenic nerve, pericardium, or brachiocephalic vein, for which dual-port thymectomy (3), trans-subxiphoid robotic thymectomy (4), or median sternotomy is performed. Despite intercostal nerve impairment caused by the insertion of the robotic arm through the intercostal space, trans-subxiphoid robotic thymectomy is particularly valuable because the camera inserted via the subxiphoid incision provides a sufficient operative field in the neck region and enables identification of both phrenic nerves, as in median sternotomy. Moreover, robot-assisted surgery provides excellent surgical manipulability, making it extremely useful.
Acknowledgements
None.
Footnote
Conflicts of Interest: The authors have no conflicts of interest to declare.
References
- Detterbeck FC, Kim AW, Zielinski M. Looking in from above and up from below: new vistas in thoracic surgery. Innovations (Phila) 2012;7:161-4. [PubMed]
- Suda T, Sugimura H, Tochii D, et al. Single-port thymectomy through an infrasternal approach. Ann Thorac Surg 2012;93:334-6. [PubMed]
- Suda T, Ashikari S, Tochii D, et al. Dual-port thymectomy using subxiphoid approach. Gen Thorac Cardiovasc Surg 2014;62:570-2. [PubMed]
- Suda T, Tochii D, Tochii S, et al. Trans-subxiphoid robotic thymectomy. Interact Cardiovasc Thorac Surg 2015;20:669-71. [PubMed]





